Scientific Reports, 02 July 2025
Background
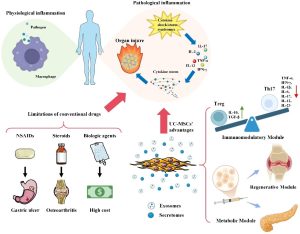
Central nervous system (CNS) injuries are a major cause of permanent neurological deficits, and current therapeutic strategies remain limited. Mesenchymal stem cells derived from Wharton’s Jelly (WJ‑MSCs) have emerged as a promising approach in neural tissue regeneration due to their paracrine trophic effects, immunomodulatory properties, and support for neurovascular remodeling.
Study Design and Methods
- Injury Model
- Adult Wistar rats were subjected to focal cortical–thalamic injury induced by ouabain, an inhibitor of Na⁺/K⁺-ATPase, to model neuronal damage and ionic imbalance.
- Stem Cell Transplantation
- Human WJ‑MSCs were cultured under:
- Normoxia (21% O₂) or
- Physioxia (5% O₂) – mimicking physiological oxygen levels.
- Transplantation was performed using:
- Phosphate-buffered saline (PBS) suspension
- or Fibrinogen-based hydrogel as a delivery matrix.
- Growth Factor Evaluation
- Expression levels of:
- BDNF (Brain-Derived Neurotrophic Factor)
- GDNF (Glial Cell Line-Derived Neurotrophic Factor)
- VEGF-A (Vascular Endothelial Growth Factor A)
- Assessed at 24 hours, days 7, 14, and 21 post-injury via:
- qPCR of the cortex–thalamus tissue
- and protein quantification in cerebrospinal fluid (CSF).
Key Results
- BDNF Expression
- In the ouabain-only (OUA) group, BDNF was strongly upregulated at 24 hours as an acute endogenous response.
- In MSC-transplanted groups, BDNF levels increased significantly from day 7 onward, peaking on day 21, particularly in the 5% O₂ + hydrogel group.
- GDNF Expression
- Initially reduced post-injury at 24 hours, GDNF levels rose from day 7, with the highest expression in 5% O₂ MSCs delivered via hydrogel.
- VEGF-A Expression
- VEGF-A decreased transiently at 24 hours but increased from day 7 onward, supporting vascular remodeling.
- Differences were minimal between oxygenation or delivery formats, although the fibrin gel alone (without MSCs) appeared to suppress VEGF-A expression.
- Protein Levels in CSF
- Protein concentrations mirrored the mRNA expression trends in brain tissue, confirming the systemic effects of transplanted WJ‑MSCs.
Scientific Insights
- WJ‑MSCs promote tissue repair and neurotrophin expression in injured brain microenvironments primarily via paracrine mechanisms.
- Physioxia (5% O₂) culture preserved the functional potency of MSCs and enhanced therapeutic efficacy.
- The hydrogel scaffold served as a critical delivery platform, improving cell retention and paracrine factor longevity at the injury site.
Conclusion and Implications
- Transplanting WJ‑MSCs cultured under physiologic oxygen levels and delivered via hydrogel significantly increased the long-term expression of BDNF and GDNF.
- These findings underscore the therapeutic potential of optimized MSC transplantation protocols for neural repair in conditions such as traumatic brain injury, stroke, and neurodegenerative diseases.
- Further investigation into delivery systems and preconditioning strategies will be essential for translating this approach into clinical neuroregenerative medicine.
References
Lech, W., Kot, M., Welniak-Kaminska, M., Drabik, M., Buzanska, L., & Zychowicz, M. (2025). Dynamic changes in BDNF, VEGF, and GDNF after transplanting human protein-based scaffolds with Wharton’s Jelly MSCs in a rat brain injury model. Scientific Reports, 15(1), 22625.
Source: Scientific Reports
Link: https://www.nature.com/articles/s41598-025-04269-w#citeas