Stem Cell Res Ther, 08/01/2026
Introduction
Myocardial infarction (MI) is characterized by obstruction of coronary perfusion and reduced oxygen supply to the myocardium, imposing a significant global medical, social, and economic burden. The widespread application of revascularization therapies has improved survival after the initial ischemic event; however, the incidence of post-MI heart failure has increased. The infarcted heart undergoes remodeling, including disproportionate thinning of necrotic myocardium and hypertrophy of non-necrotic myocardium, leading to post-MI heart failure. Current pharmacological treatments, cardiac assist devices, and cardiac resynchronization therapy can slow cardiac functional decline, reverse heart failure progression, and reduce mortality, but cannot replace lost cardiomyocytes. Regenerative therapy using exogenous cells to regenerate and repair the ischemic heart is a promising strategy in cardiovascular disease. The main goal of cell-based therapies is to directly regenerate myocardium with exogenous cells or activate endogenous repair mechanisms through paracrine effects. Nevertheless, immune rejection remains an unavoidable barrier limiting the clinical application of these regenerative therapies.
Existing strategies to achieve immune tolerance include induction of central and peripheral immune tolerance, use of immunosuppressive drugs, bioengineered cellular grafts, and generation of “universal” cells. Recent studies suggest that establishing peripheral immune tolerance is a promising approach to enhance cell retention. Our previous studies demonstrated that intravenous administration of human mesenchymal stem cells (hMSCs) improves engraftment and therapeutic efficacy of intramyocardially and intramuscularly transplanted cells in MI and hindlimb ischemia models. However, high-dose intravenous MSC injection does not further increase immunomodulatory efficacy. In addition, human leukocyte antigen G (HLA-G) has been reported to exert immunomodulatory effects by inducing HLA-G+ and Foxp3+ regulatory T cells and modulating natural killer (NK) cell activity. HLA-G is a key molecule in maternal–fetal immune tolerance and has been widely discussed in solid organ transplantation. However, whether upregulation of HLA-G expression can further improve survival and therapeutic efficacy of cell therapy remains unclear.
In our recent work, human umbilical cord blood–derived MSCs overexpressing HLA-G1 were successfully generated. Therefore, this study hypothesizes that, compared with conventional hUCB-MSCs, systemic intravenous administration of HLA-G1–overexpressing hUCB-MSCs can enhance cell retention and improve the therapeutic efficacy of intramyocardially transplanted human induced pluripotent stem cell–derived cardiomyocytes (hiPSC-CMs) in a mouse MI model. Meanwhile, the underlying mechanisms of the immunomodulatory effects of HLA-G1–overexpressing MSCs were also investigated.
Methods
Culture and preparation of hUCB-MSCs and hiPSC-CMs
hUCB-MSCs were purified, characterized by specific markers, selected for multilineage differentiation potential, and used at passages 4–6; they were then administered intravenously in mice. Beating hiPSC-CMs were confirmed for cardiomyocyte characteristics by immunostaining and flow cytometry (≈95% cTnT positive) and labeled with DiR prior to intramyocardial transplantation.
Human HLA-G1 transduction
hUCB-MSCs were transduced with lentivirus with or without the HLA-G1 gene to generate an overexpressing line. The cells were divided into three groups: native MSCs, MSC-NC (control GFP-expressing lentivirus), and MSC-HLA-G1 (HLA-G1–overexpressing lentivirus). Transduction efficiency was confirmed after culture by qRT-PCR and western blot, demonstrating successful upregulation of HLA-G1.
Establishment of mouse MI model, grouping, and cell tracing
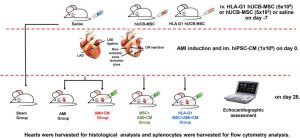
ICR mice were used to evaluate the anti-inflammatory effects of MSCs. One week before induction of acute myocardial infarction (AMI), mice received intravenous injection of saline, hUCB-MSCs, or HLA-G1–overexpressing hUCB-MSCs. AMI was induced under anesthesia by ligation of the left anterior descending coronary artery; electrocardiography was used to confirm successful model establishment. After 30 minutes, mice received intramyocardial injection of culture medium or DiR-labeled hiPSC-CMs at the peri-infarct border zone; antibiotics were administered to prevent infection. Animals that died during the procedure were excluded from analysis.
A total of 30 mice were randomly divided into five groups: Sham, AMI, AMI + CM, MSC + AMI + CM, and HLA-G1 MSC + AMI + CM. After 28 days, cardiac function was assessed by echocardiography, and animals were then euthanized by anesthetic overdose to collect hearts and organs for tumor formation evaluation. Cell retention was monitored on days 7 and 28 using an IVIS fluorescence imaging system based on the signal from DiR-labeled hiPSC-CMs.
Echocardiographic examination
On day 28, cardiac function of AMI mice was evaluated by transthoracic echocardiography using a FUJIFILM VisualSonics system with a 10 S-RS transducer. Mice were anesthetized with inhaled isoflurane and maintained at a heart rate of 500–600 bpm. Measurements were performed by an experienced technician in accordance with the recommendations of the European Society of Cardiology. Mice were placed in the left lateral decubitus position, parasternal views of the left ventricle were obtained, and the long-axis view was used for cardiac function analysis.
Cardiac function was assessed using standard M-mode parameters including left ventricular end-systolic diameter (LVESD), left ventricular end-diastolic diameter (LVEDD), fractional shortening (FS), and left ventricular ejection fraction (LVEF). The average of three cardiac cycles per animal was calculated by an investigator blinded to the experiment.
Histological assessment
After euthanasia, hearts were harvested for histological evaluation. Tissues were fixed in 4% buffered formalin, dehydrated through graded ethanol, paraffin-embedded, and sectioned at 5 µm thickness. H&E staining was performed using a commercial kit. Masson trichrome staining was used to evaluate infarct size; infarct area was calculated as the ratio of blue-stained fibrotic tissue to total left ventricular area, measured on three sections and averaged.
Apoptosis in the peri-infarct region was assessed by TUNEL staining. Neovascularization was examined by immunofluorescence staining for α-SMA and vWF, and vessel density was calculated as the number of positive vessels per mm². Data were collected from six random high-power fields (40×) across three sections using a blinded evaluation protocol and recorded with an automated microscopy system.
Flow cytometry analysis
Splenocytes were harvested after euthanasia for flow cytometric analysis. After red blood cell lysis, single-cell suspensions were prepared in PBS. Approximately 1 × 10⁶ cells were incubated with anti-CD16/32 blocking antibody and 7AAD in flow buffer.
T cell and NK cell populations were stained with specific antibodies to identify CD3+, CD4+, CD8+ T cells and NK cells. Regulatory T cells (Tregs) were stained using a dedicated kit including CD4, CD25, and Foxp3. Samples were analyzed on a CytoFLEX flow cytometer, with fluorescence minus one (FMO) control used for multicolor gating strategy, and data were processed using FlowJo software.
The changes in cytokines
Splenic CD4+ cells were co-cultured in vitro with hUCB-MSCs or HLA-G1–overexpressing hUCB-MSCs to evaluate immunomodulatory effects. CD4+ cells were isolated by anti-CD4-FITC antibody staining and sorted by FACS. Subsequently, 5 × 10⁵ CD4+ cells were co-cultured with 1 × 10⁵ MSCs in a transwell system for 7 days. Culture supernatants were collected for cytokine quantification using a multiplex assay kit according to the manufacturer’s instructions.
Statistical analysis
Data were analyzed using SPSS 27.0 and presented as mean ± SEM. Multiple group comparisons were performed using one-way ANOVA followed by Tukey’s test; p < 0.05 was considered statistically significant.
Results
Thirty-five mice underwent myocardial infarction induction, of which five died, leaving 30 mice included in the analysis and randomly assigned to five groups. hUCB-MSCs expressed characteristic MSC markers and lacked hematopoietic markers. Lentiviral transduction was successful, and the MSC-HLA-G1 group showed markedly increased HLA-G1 expression compared with control groups.
hiPSC-CMs were confirmed to possess cardiomyocyte characteristics by immunostaining and flow cytometry (~95% cTnT positive). No tumor formation was observed at injection sites or in organs. Electrocardiography showed ST-segment elevation, confirming successful establishment of the myocardial infarction model.
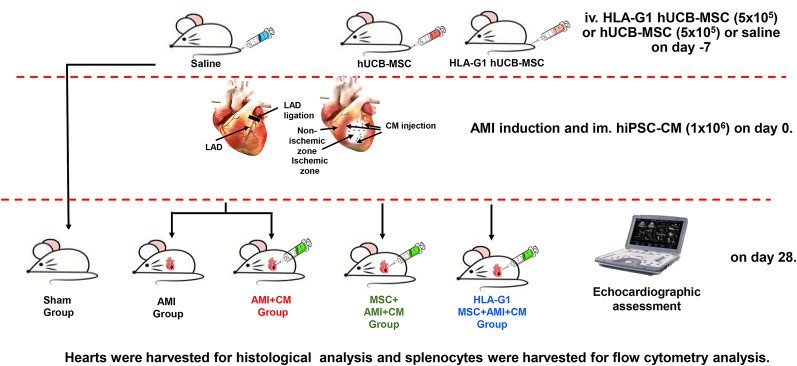
Figure 1. Flow chart of the experiment
The Immunomodulatory effect of hUCB-MSCs and HLA-G1 overexpressing hUCB-MSCs
Flow cytometric analysis of splenocytes showed no differences among the Sham, AMI, and AMI + CM groups. However, intravenous administration of hUCB-MSCs or HLA-G1–overexpressing hUCB-MSCs significantly increased the proportion of splenic Tregs compared with the AMI group, with a greater increase observed in the MSC-HLA-G1 group than in conventional MSCs. In contrast, AMI increased splenic NK cells, and transplantation of hiPSC-CMs did not affect this parameter; MSC administration reduced NK cells, with MSC-HLA-G1 producing a stronger reduction. No significant changes were observed in the CD4+ T cell population.
In the in vitro co-culture experiment, hUCB-MSCs reduced pro-inflammatory cytokines (IFN-γ, TNF-α, IL-2, IL-17A) without altering anti-inflammatory cytokines; suppression of pro-inflammatory cytokines was more pronounced with HLA-G1–overexpressing MSCs. These results indicate that HLA-G1–overexpressing MSCs exert stronger immunomodulatory effects than conventional MSCs, at least partly through reducing pro-inflammatory cytokine secretion.
Increased engraftment and cell retention after transplantation
Fluorescence imaging on days 7 and 28 showed that intravenous administration of hUCB-MSCs or HLA-G1–overexpressing hUCB-MSCs significantly increased engraftment and retention of hiPSC-CMs at the peri-infarct region compared with the cell transplantation–only group. This effect was more pronounced in the HLA-G1–overexpressing MSC group than in conventional MSCs and was sustained through day 28.
These results indicate that intravenously delivered MSCs improve survival of intramyocardially transplanted hiPSC-CMs, consistent with previous studies. HLA-G1–overexpressing MSCs produced a stronger effect, suggesting that enhanced immunomodulation contributes to improved graft retention.
Decreased infarct size and apoptosis rate after transplantation
H&E, Masson, and TUNEL staining showed that the AMI group had a large infarct area and a high rate of apoptosis. Transplantation of hiPSC-CMs reduced infarct size and apoptosis in the peri-infarct region compared with the AMI group; this effect was enhanced when combined with intravenous administration of hUCB-MSCs.
Notably, HLA-G1–overexpressing hUCB-MSCs further reduced infarct size and apoptosis, indicating improved therapeutic efficacy, consistent with the stronger immunomodulatory role of HLA-G1.
Increased angiogenesis and neovascularization
Immunofluorescence staining for α-SMA and vWF showed that AMI reduced vessel density in the peri-infarct region. Transplantation of hiPSC-CMs improved angiogenesis, and this effect was further enhanced when combined with intravenous administration of hUCB-MSCs. Notably, HLA-G1–overexpressing MSCs produced an even greater increase in angiogenesis and neovascularization.
These findings indicate that intravenously delivered MSCs enhance the therapeutic efficacy of hiPSC-CMs, with HLA-G1–overexpressing MSCs exerting superior pro-angiogenic effects.
Improved LV systolic function after transplantation
Echocardiography showed that AMI severely impaired left ventricular function (reduced LVEF and FS, and increased LVEDD and LVESD). Transplantation of hiPSC-CMs markedly improved cardiac functional parameters; this effect was further enhanced when combined with intravenous administration of hUCB-MSCs.
Notably, HLA-G1–overexpressing hUCB-MSCs improved cardiac function more strongly than conventional MSCs, with higher LVEF and FS and a greater reduction in LVESD. These results indicate that HLA-G1–overexpressing MSCs enhance recovery of cardiac function after myocardial infarction.
Discussion
The results show that intravenous administration of hUCB-MSCs significantly increased engraftment of intramyocardially transplanted hiPSC-CMs, consistent with previous studies. HLA-G1–overexpressing hUCB-MSCs exhibited stronger immunomodulatory effects than conventional MSCs. Reduction of pro-inflammatory cytokines and increased splenic Tregs improved immune regulation and enhanced graft retention, which was associated with recovery of cardiac function, accompanied by increased angiogenesis and reduced inflammation and apoptosis in the peri-infarct region.
Immunomodulatory effects of HLA-G1 overexpressing hUCB-MSCs
Our previous studies showed that intravenous injection of 5 × 10⁵ MSCs significantly increased engraftment and therapeutic efficacy of intramyocardially transplanted hiPSC-CMs, whereas increasing the MSC dose did not further enhance immunomodulatory effects. HLA-G is a non-classical class I HLA molecule that plays an important role in maternal–fetal immune tolerance and is often used to reduce inflammation and enhance immunomodulatory efficacy in cell transplantation. Compared with MSCs from other sources, hUCB-MSCs express higher levels of HLA-G1 under standard culture conditions. In this study, HLA-G1–overexpressing hUCB-MSCs were used to improve engraftment of hiPSC-CMs. Because splenic Tregs peak 7 days after MSC infusion, MSCs were administered intravenously 7 days before transplantation. The results showed that HLA-G1–overexpressing MSCs significantly improved engraftment, associated with increased splenic Tregs and reduced NK cells, suggesting attenuation of rejection through both MHC-dependent and MHC-independent mechanisms.
To further evaluate immunomodulatory effects, cytokines were analyzed after 7 days of co-culture of MSCs with CD4⁺ splenocytes. In addition to IFN-γ, TNF-α, and IL-17A, MSCs also reduced the pro-inflammatory cytokine IL-2. Although low IL-2 levels are typically associated with reduced Tregs, Tregs were not decreased in this study, possibly because MSCs secrete factors such as IDO and HLA-G that promote Treg expansion. Both hiPSC-MSCs and hUCB-MSCs showed similar immunomodulatory efficacy, but it cannot yet be concluded which cell type is superior.
Overall, intravenous administration of hUCB-MSCs modulates immune responses by reducing pro-inflammatory cytokines and NK cells while increasing Tregs, thereby improving engraftment of intramyocardially transplanted hiPSC-CMs.
Mechanism of improved cardiac function
The results showed that intravenous administration of hUCB-MSCs or HLA-G1–overexpressing hUCB-MSCs markedly improved cardiac function 28 days after myocardial infarction, with greater efficacy observed in the HLA-G1 group. Therapeutic benefits were associated with reduced infarct size, decreased apoptosis, enhanced graft retention, and increased neovascularization. The primary mechanisms include:
- Systemic immunomodulatory effects of intravenously delivered MSCs
- Paracrine effects of intramyocardially transplanted hiPSC-CMs, rather than direct myocardial regeneration.
No tumor formation or arrhythmias related to cell transplantation were observed, confirming the safety of the therapy. Upregulation of HLA-G1 in MSCs further enhanced myocardial repair efficacy. This study reinforces the clinical potential of combining systemic MSC infusion with cardiomyocyte transplantation to restore cardiac function after myocardial infarction.
Limitation
This study has several limitations. First, MSCs were administered intravenously 7 days before myocardial infarction based on prior data showing that Tregs peak after 7 days, but the small sample size and inability to fully replicate clinical conditions limit translation; therefore, the optimal timing of MSC administration in practice requires further investigation. Second, although repeated MSC injections may be more effective than a single dose, it remains unclear whether periodic infusion of hUCB-MSCs every 7 days would further enhance immunomodulatory effects; however, HLA-G1 overexpression clearly improved these effects. Third, because mice lack a human HLA-G homolog, the underlying signaling pathways could not be fully elucidated, although modulation of NK cells and Tregs was confirmed. Fourth, it is difficult to assess whether infusion of HLA-G1–overexpressing MSCs increases systemic HLA-G levels in the mouse model, but results showed a significant reduction in pro-inflammatory cytokines in splenic CD4+ cells. Fifth, late or transient arrhythmias cannot be excluded because monitoring was performed only on days 7 and 28; safety requires further preclinical validation. Finally, therapeutic efficacy was evaluated only through day 28, so long-term durability remains to be determined.
Conclusion
In summary, this study demonstrates that hUCB-MSCs overexpressing the HLA-G1 gene exhibit superior immunomodulatory efficacy. Intravenous administration of HLA-G1–overexpressing hUCB-MSCs enhanced engraftment of intramyocardially transplanted hiPSC-CMs and improved cardiac function by increasing systemic Tregs, reducing systemic NK cells, and modulating immune cytokine release. Future studies are needed to determine the optimal delivery route of hUCB-MSCs before clinical translation.
References
Zhu W, Kong J, Li HX, Jiang TB, Sun SJ, Zou C (2026). Immunomodulatory effect of mesenchymal stromal cell overexpressing HLA-G1 in cell-based therapy for myocardial infarction. Stem Cell Res Ther. Jan 8;17(1):73.
Nguồn: Stem Cell Res Ther
Link: https://link.springer.com/article/10.1186/s13287-026-04897-7