BMC Musculoskelet Disord, 29/12/2025
Introduction
Knee osteoarthritis (KOA) is a chronic degenerative disorder with a high and rapidly increasing prevalence due to population aging, leading to persistent pain, joint deformity, and substantial economic burden. Currently, no therapy has been recognized as disease-modifying, and many patients with advanced disease ultimately require joint replacement, although surgery does not fully restore sports participation. Therefore, developing biological treatments capable of targeting the underlying pathophysiology of KOA remains a major clinical priority.
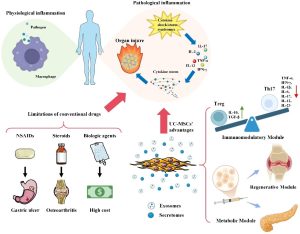
Mesenchymal stem cell (MSC)–based therapy has emerged as a promising approach owing to its immunomodulatory, anti-inflammatory, and chondroprotective properties. The therapeutic effects of MSCs are attributed not only to their differentiation capacity but also to paracrine mechanisms mediated by cytokines and exosomes. Preclinical studies suggest that MSCs can slow cartilage degeneration, suppress joint inflammation, and promote tissue regeneration.
Among MSC sources, umbilical cord–derived mesenchymal stem cells (UC-MSCs) are highly valued because of their strong proliferative capacity, low immunogenicity, noninvasive procurement, and minimal ethical concerns. Recent meta-analyses indicate that UC-MSCs may provide superior pain relief and functional improvement compared with other MSC sources. However, most available evidence comes from small-scale studies, underscoring the need for high-quality randomized clinical trials.
Accordingly, this study was designed as a randomized, double-blinded, controlled trial to rigorously evaluate the safety and efficacy of UC-MSCs in the treatment of knee osteoarthritis.
Materials and Methods
Study design
The study was conducted at Wuhan Union Hospital between January 2021 and June 2022. It was registered as a clinical trial and approved by the institutional ethics committee in accordance with the Declaration of Helsinki (approval No. 2019SC02). The trial was designed as a randomized, double-blinded, placebo-controlled study to evaluate the safety and efficacy of intra-articular UC-MSC injection in patients with knee osteoarthritis.
Patient
The study population included patients aged 40–75 years with unilateral knee osteoarthritis of Kellgren–Lawrence grade 1–3 lasting more than 6 months and unresponsive to conservative treatment. Exclusion criteria comprised other severe musculoskeletal diseases, significant limb malalignment, major ligament or meniscal injury, intra-articular interventions within the previous year, infection, malignancy, current use of immunosuppressive or anticoagulant therapy, body mass index ≥ 30, or any condition that could interfere with clinical evaluation. All participants provided written informed consent.
UC-MSCs isolation and culture
The use of human umbilical cord tissue was ethically approved, and informed consent was obtained from donors. Umbilical cord–derived mesenchymal stem cells (UC-MSCs) were isolated, expanded, and characterized according to standardized procedures to meet MSC criteria. Umbilical cord tissue was minced, processed under sterile conditions, and cultured in serum-free medium until high confluence was reached, followed by passaging; fifth-passage cells were used for the trial. Cell manufacturing complied with Good Manufacturing Practice (GMP) standards, with each therapeutic dose containing approximately 2 × 10⁷ cells. Prior to administration, cells were tested for microbial contamination, mycoplasma, and endotoxin, then suspended in saline supplemented with human albumin for intra-articular injection.
Intervention
Eligible participants were randomly assigned in a 1:1:1 ratio to receive placebo, hyaluronic acid (HA), or UC-MSCs according to a computer-generated allocation schedule. Each patient received a single intra-articular injection at baseline under ultrasound guidance by a physician not involved in outcome assessment. The study was double-blinded, with both participants and evaluating physicians unaware of treatment allocation. The UC-MSC dose consisted of 2 × 10⁷ cells suspended in 2.5 mL of 0.9% saline supplemented with 1% human albumin; the HA group received 2.5 mL sodium hyaluronate; and the placebo group received an equivalent volume of vehicle solution without cells. Participants were followed for 24 weeks, with visits at weeks 2, 6, 12, and 24 conducted by an independent blinded physician.
Outcomes
The primary outcome was the safety of UC-MSCs, assessed through clinical examination, laboratory testing, electrocardiography, and documentation of adverse events at each follow-up visit. Adverse events were classified using the WHO-UMC causality assessment system and graded for severity according to the NCI-CTCAE scale.
Secondary outcomes evaluated therapeutic efficacy based on clinical and imaging measures. Clinical assessments included VAS for pain, WOMAC and KSS for knee joint function, and EQ-5D-5L for quality of life. MRI findings were analyzed using the MOAKS scoring system to monitor disease progression, focusing on cartilage lesions, synovitis, and bone marrow lesions. All data were collected at baseline and at weeks 2, 6, 12, and 24 by physicians blinded to treatment allocation.
Sample size
This was an exploratory randomized trial designed to provide preliminary evidence on safety and efficacy prior to conducting a large-scale RCT. The study required 18 participants per group, for a total of 54 participants.
Statistical analysis
The prespecified analyses were performed on the intent-to-treat (ITT)..
Results
Patient characteristics
Of the 69 patients screened, 55 met the eligibility criteria and were randomly assigned to the three groups. After excluding withdrawals and patients lost to follow-up, 15 patients in the placebo group, 16 in the HA group, and 17 in the UC-MSC group completed the study. The groups were comparable in baseline demographic and clinical characteristics.
Safety outcomes
No serious adverse events, permanent disability, deaths, septic arthritis, or neoplasms were reported during the study. Adverse events occurred in 18.8% of patients in the HA group and 29.4% in the UC-MSC group, most commonly injection-site pain, with no statistically significant difference between groups. Symptoms were transient, responded to rest or acetaminophen, and resolved within 2 days. One patient in the UC-MSC group developed a mild fever after injection that resolved spontaneously. All adverse events were mild to moderate in severity, and no participant discontinued the study due to safety concerns.
Clinical outcomes
VAS, WOMAC, KSS, and EQ-5D scores showed no significant differences among the three groups at 6 months compared with baseline. However, in the subgroup of patients with synovitis identified on baseline MRI, only the UC-MSC group demonstrated a significant improvement in synovitis at 24 weeks, whereas the placebo and HA groups showed no meaningful change. MRI images revealed a gradual reduction in synovial thickening and edema following UC-MSC injection.
Among patients with synovitis, the placebo group showed no significant changes in VAS, KSS, WOMAC, or EQ-5D scores. The HA group exhibited a transient increase in pain and symptoms during the first 2 weeks, followed by gradual improvement. The UC-MSC group showed greater pain reduction compared with the other two groups, although this difference did not reach statistical significance; however, WOMAC scores improved significantly at the final follow-up (P = 0.036). Quality of life measured by EQ-5D also improved significantly at later follow-up time points.
When analyzing patients with synovitis and baseline pain severity of VAS ≥ 4, neither the placebo nor HA groups demonstrated significant improvement in clinical scores. In contrast, the UC-MSC group showed marked pain reduction at weeks 6 and 24 compared with baseline, along with a significant improvement in quality of life (EQ-5D) at week 24. WOMAC scores tended to improve over the long term but were not consistently sustained during the early phase.
Radiological outcomes
No new cyst formation, tumors, or heterotopic ossification were observed in bone, cartilage, vasculature, or synovium. After 6 months, MRI evaluation using the Global MOAKS system showed no significant differences among groups in the size of articular cartilage defects or bone marrow lesions.
Discussion
This study demonstrates that intra-articular injection of umbilical cord–derived mesenchymal stem cells (UC-MSCs) is safe and well tolerated in the treatment of knee osteoarthritis, and provides meaningful clinical improvement in patients with concomitant synovitis, particularly those with moderate-to-severe pain. No serious adverse events were observed during the 6-month follow-up; reported reactions were mainly transient injection-site pain.
Knee osteoarthritis is a multifactorial disease in which inflammation plays a key role in driving cartilage degeneration and disease progression. Owing to their strong immunomodulatory and anti-inflammatory properties, MSCs — especially UC-MSCs — are considered a promising biological therapy. UC-MSCs offer advantages in proliferative capacity, low immunogenicity, and abundant availability, making them suitable for clinical application. The present findings show that UC-MSCs improve synovitis on MRI, an important observation given the close association between synovial inflammation, pain, and functional impairment. In patients with baseline VAS pain ≥ 4, UC-MSC treatment produced clear improvements in pain and function compared with placebo and HA, while also enhancing quality of life.
The absence of clear structural cartilage changes on MRI after 6 months may be attributable to the short follow-up duration and limited sample size. Previous preclinical data and longer-term studies suggest that cartilage regenerative effects of MSC therapy may require a longer period to become radiologically detectable.
This study has several limitations, including small sample size, short follow-up, and lack of comparison with a broader range of treatment options. Nevertheless, these preliminary results strengthen the evidence that UC-MSCs have potential as a disease-modifying therapy for knee osteoarthritis, particularly in patients with a prominent inflammatory component. Larger trials with extended follow-up are required to confirm long-term efficacy and safety.
Conclusion
This trial shows that umbilical cord–derived mesenchymal stem cell (UC-MSC) therapy is safe and well tolerated in the treatment of knee osteoarthritis, with a safety profile comparable to placebo and HA. Intra-articular UC-MSC injection provides clear pain relief and functional improvement in patients with synovitis, particularly those with moderate-to-severe pain. However, the small sample size and the 6-month follow-up period represent important limitations. Large multicenter trials with long-term follow-up are required to confirm and extend these findings. In addition, as MSC therapy is a novel intervention, future studies should incorporate cost-effectiveness analyses to clarify its economic value compared with standard treatment.
References
Tong W, Shi Y, Wu D, Jia J, Xiang E, Xu D, Rao W, Hu Q, Liao Q, Xiao C, Zheng S, Xu W, Tian H (2025). Intra-articular injection of umbilical cord-derived mesenchymal stem cells is safe and effective for moderate to severe knee osteoarthritis with synovitis: a double blinded and randomized controlled trial. BMC Musculoskelet Disord. Dec 29;27(1):84.
Nguồn: BMC Musculoskelet Disord