Diseases, 08/01/2026
Introduction
In patients undergoing median sternotomy, particularly after coronary artery bypass grafting (CABG), deep sternal wound infection (DSWI) may progress to chronic sternal osteomyelitis—a serious clinical complication. The incidence of DSWI following sternotomy ranges from 0.25–5%. Despite timely and comprehensive treatment, mortality remains alarmingly high; systematic reviews and meta-analyses indicate that DSWI may account for up to approximately half of in-hospital deaths, making it one of the most life-threatening complications of cardiac surgery.
Mesenchymal stem cells (MSCs) are considered a promising approach for the treatment of chronic infections, including DSWI. The MSC secretome exhibits notable antimicrobial properties through the secretion of antimicrobial peptides such as LL-37—which can directly disrupt bacterial membranes—while also exerting potent immunomodulatory effects by “reprogramming” local immune cells, particularly macrophages. This activation promotes phenotypic switching from pro-inflammatory M1 to anti-inflammatory and phagocytic M2 macrophages, thereby enhancing bacterial clearance and limiting chronic tissue-destructive inflammation. In addition, MSC-derived extracts demonstrate high regenerative potential in skin wound healing through multiple mechanisms, including reactivation of dermal fibroblasts. MSCs themselves can differentiate into connective tissue, replace damaged cells at the wound site, and promote angiogenesis as well as cell migration to the injured area.
However, not all MSC sources exhibit equivalent efficacy and regenerative capacity. Perinatal MSCs (derived from tissues surrounding the fetus) offer several advantages over adult MSCs, including a more “primitive” phenotype, greater proliferative capacity, and lower immunogenicity. Notably, they demonstrate low tumorigenic potential or even exhibit anti-tumor activity. Furthermore, these MSCs are obtained from biological materials typically discarded after birth, such as amniotic fluid, placenta, and umbilical cord, and therefore are associated with minimal ethical concerns.
Wharton’s jelly—a component of the umbilical cord—is a rich source of immature MSCs. Wharton’s jelly-derived MSCs (WJ-MSCs) are highly regarded in regenerative medicine due to their strong paracrine regulatory capacity through the secretion of numerous bioactive factors. WJ-MSCs have been shown to promote epithelial proliferation and accelerate wound healing, with documented efficacy in deep skin wounds and infected wounds following severe burns. However, direct clinical application of WJ-MSCs remains limited, and data are still lacking regarding their regenerative efficacy, particularly in chronic infected surgical wounds in elderly patients—a population characterized by impaired tissue repair capacity.
In this report, we present a case of an elderly male patient diagnosed with refractory chronic sternal osteomyelitis and mediastinitis following CABG. The persistence of a non-healing chronic wound, compounded by perioperative COVID-19 infection, necessitated a personalized, multi-stage treatment strategy to optimize clinical outcomes.
Case Presentation
A 63-year-old male patient experienced his first acute myocardial infarction in 2014 and underwent coronary stent placement according to a staged interventional strategy. In 2016, he developed recurrent severe angina. Follow-up coronary angiography revealed multivessel coronary artery disease. In December 2016, the patient underwent off-pump coronary artery bypass grafting (CABG). The procedure included left internal mammary artery–left anterior descending artery grafting, along with autologous great saphenous vein (vena saphena magna) grafts to the diagonal branch, obtuse marginal branch of the circumflex artery, and the circumflex artery.
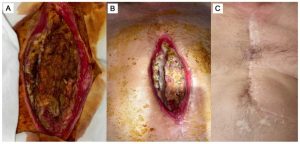
Approximately three weeks after CABG (early 2017), the patient developed purulent discharge at the lower third of the sternotomy scar (Figure 1A). In 2017, 2018, and 2020, he underwent three additional surgical excisions of the fistulous tract. However, persistent inflammation, recurrent fistula formation, and delayed wound healing continued despite antibiotic therapy guided by susceptibility testing for Staphylococcus aureus infection (≈10⁵ CFU/mL), initially treated with Cefuroxime (0.75 g every 8 hours), followed by Ceftriaxone (2 g every 12 hours).
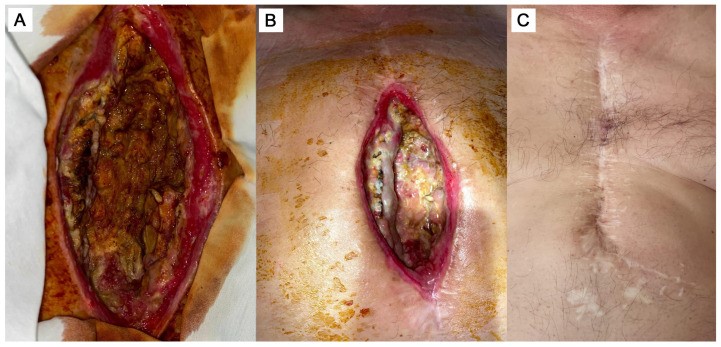
Figure 1. The state of the surgical wound before (A), 6 days after the commencing of cell product administration as an additive to the surgical correction (B), and 4 years after the treatment (C).
On May 11, 2021, the patient underwent reoperation due to a marked increase in systemic inflammatory response (CRP = 168.46 mg/L; presepsin = 351 pg/mL; leukocytes = 18.7 × 10⁹/L). The fistulous tract, which penetrated the sternum and extended into the pericardial cavity and right ventricle, was excised. However, over the following two weeks, poor wound healing and signs of chronic inflammation persisted.
On May 27, 2021, after providing informed consent for treatment, the patient received perilesional infiltration of an allogeneic cell preparation containing MSCs and fibroblasts. The cell source was obtained from umbilical cord tissue provided by an obstetric facility (not a commercial product). The preparation had a concentration of 1 × 10⁶ cells/mL in 0.9% NaCl solution. The injection procedure was performed using a sterile 1.0 mL insulin syringe with a fine needle to ensure accurate dosing and minimize tissue trauma. The solution was administered intradermally and subcutaneously along the wound periphery, 0.5–1.0 cm from the wound edge. The procedure was repeated three times at 48-hour intervals (days 1, 3, and 5) to maintain therapeutic efficacy.
The injection technique involved creating dermal papules by administering 0.1–0.2 mL per injection point, spaced 1 cm apart, totaling 10–15 injections around the fistulous tract to stimulate angiogenesis and promote epithelialization of the wound margins prior to planned chest wall reconstruction.
In the subsequent days, marked local improvement was observed, including cessation of pathological discharge and progressive wound healing (Figure 1B).
During preparation for the next surgery, screening detected SARS-CoV-2 RNA; the patient remained asymptomatic, and this finding did not significantly affect wound healing. Before and after reconstructive surgery, the patient received systemic antibiotic therapy with piperacillin + tazobactam (4.5 g every 8 hours).
On July 12, 2021, local examination revealed that the wound remained open; systemic inflammatory markers were still elevated (CRP = 130.88 mg/L; erythrocyte sedimentation rate = 63 mm/h). Echocardiography showed a reduced left ventricular ejection fraction (LVEF) of 49–50% with signs of left ventricular aneurysm formation.
On July 16, 2021, the patient underwent definitive reconstructive surgery, including sternal fixation with metal plates, autologous iliac crest bone grafting, and reattachment of the ribs to the sternum. At the time of intervention, the wound bed exhibited healthy granulation tissue with no pathological discharge. The sternal edges and ribs 5–7 were mobilized to facilitate structural reconstruction; the autologous bone graft was secured to the sternal defect using two metal plates and anchored to the costochondral junctions of the corresponding ribs.
Postoperatively, inflammatory markers gradually decreased (CRP reduced to 47.31 mg/L on July 27, 2021). The retrosternal drain was removed on July 19, 2021. Follow-up echocardiography on July 22, 2021 showed a further decrease in LVEF to 39%; however, the patient remained hemodynamically stable, without congestion or peripheral edema. Heart failure was effectively managed according to standard protocol with beta-blocker (bisoprolol), angiotensin-converting enzyme inhibitor (lisinopril), and aldosterone antagonist therapy. The patient was discharged on August 2, 2021, after 21 days of inpatient treatment, in stable condition.
After discharge, the patient was followed on an outpatient basis. The sternum was stable, and the wound achieved primary healing. General condition improved, with marked reduction in local inflammatory signs; leukocyte count and CRP gradually approached near-normal levels (after 1 month: CRP = 15.15 mg/L; leukocytes = 10.66 × 10⁹/L). During long-term follow-up, the wound closed completely with full tissue regeneration. Figure 1C illustrates the surgical site approximately four years after cell therapy.
Discussion
Experimental findings over the past decade have shown that human perinatal tissues such as the placenta, amniotic membrane, and umbilical cord, as well as perinatal fluids including amniotic fluid and cord blood, contain multiple populations of multipotent progenitor cells. These cells are collectively referred to as perinatal mesenchymal stem cells (P-MSCs) and hold particular significance in regenerative medicine due to several advantages over “adult” MSCs (A-MSCs), such as those derived from bone marrow (BM-MSCs) or adipose tissue (AD-MSCs).
First, P-MSCs possess high regenerative potential due to their more primitive phenotype and greater stemness. This is especially important in clinical applications, where therapeutic efficacy often requires cell numbers reaching hundreds of millions.
Second, P-MSCs exhibit low immunogenicity; numerous successful allogeneic transplantations without rejection have been reported. They express very low levels of major histocompatibility complex class I (MHC I), virtually no MHC class II, and lack co-stimulatory molecules such as CD80 and CD86. This “immune-privileged” property allows their use in allogeneic settings without strict human leukocyte antigen (HLA) matching requirements.
Third, P-MSCs have low tumorigenic risk and may even display anti-tumor properties. Compared with A-MSCs, they accumulate fewer genetic mutations, thereby reducing the risk of malignant transformation during in vitro expansion.
Finally, perinatal tissues such as the umbilical cord and placenta are typically discarded after birth as medical waste. Therefore, procurement of this cell source is safe, readily accessible, and non-invasive to the donor. Unlike allogeneic MSCs from adult sources, which require invasive harvesting procedures, or embryonic stem cells associated with ethical controversy, collection of perinatal tissues raises minimal ethical concerns.
Mesenchymal stem cells derived from Wharton’s jelly (WJ-MSCs) represent a subgroup of MSCs originating from perinatal tissue. Wharton’s jelly is a specialized connective tissue surrounding the umbilical cord vessels, providing mechanical protection (Figure 2A), while also serving as an important stem cell reservoir and being considered a reference source of P-MSCs.
When cultured in vitro, WJ-MSCs display a typical fibroblast-like morphology (Figure 2B) and express surface markers consistent with the criteria established by the International Society for Cell & Gene Therapy (ISCT): positive for CD90, CD105, and CD73; negative for hematopoietic markers such as CD45 and CD34.
WJ-MSCs exhibit a high proliferation rate under culture conditions, enabling the generation of therapeutically relevant cell numbers from a single donor sample within a relatively short period. Regarding differentiation potential, they are capable of in vitro differentiation into chondrocytes, osteocytes, and adipocytes.
Their immunomodulatory capacity is considered the most prominent therapeutic advantage of WJ-MSCs. Beyond their “immune-privileged” status, they actively regulate immune responses by inhibiting T-cell proliferation and modulating the activity of B cells, dendritic cells, and natural killer (NK) cells, thereby creating a strongly anti-inflammatory microenvironment.
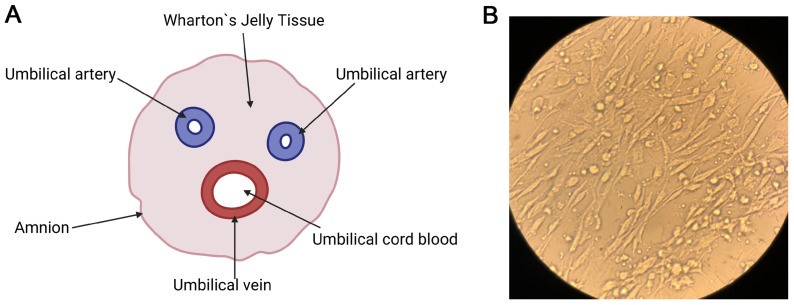
Figure 2. Mesenchymal stem cells obtained from Wharton’s jelly (WJ-MSCs). (A) Schematic representation of the human umbilical cord cross-section with main anatomical structures: the external amniotic membrane (amnion), which limits the Wharton’s jelly tissue (the mucous connective tissue), the vascular component includes two umbilical arteries (umbilical artery), and one umbilical vein (umbilical vein) containing umbilical cord blood. (B) The morphology of the WJ-MSCs cultured in vitro.
The clinical translation of MSC therapy for chronic wounds, bone infections, or chronic pain has generally produced inconsistent results. A large double-blind randomized clinical trial involving 114 patients with chronic low back pain (comparing allogeneic BM-MSCs with placebo) failed to meet its primary endpoint after 12 months of follow-up. A review of 449 MSC trials in orthopedics (up to December 2023) found that only 12.5% had published peer-reviewed results; more than half of the protocols did not clearly specify the injected cell dose. These methodological inconsistencies hinder comparability, reproducibility, and the development of MSCs into standardized therapeutic products.
Nevertheless, a meta-analysis of more than 30 studies involving approximately 2,500 patients confirmed the effectiveness of MSCs in wound closure, tissue regeneration, and angiogenesis, predominantly using adult-source MSCs (bone marrow and adipose tissue).
Adult MSCs, particularly BM-MSCs, present several limitations. Bone marrow harvesting from the iliac crest is an invasive procedure associated with pain and potential infection risk. More importantly, the frequency of MSCs in bone marrow is extremely low (0.001–0.01% of mononuclear cells), corresponding to approximately 60–600 cells/mL of aspirate, making direct clinical use impractical and necessitating prolonged ex vivo expansion, which leads to in vitro senescence. In addition, biological aging in vivo represents a fundamental limitation: the proliferative and osteogenic capacities of BM-MSCs decline markedly with age; in individuals over 60 years, alkaline phosphatase mRNA expression (a key osteogenic marker) is reduced threefold compared with those under 50 years. Consequently, a therapeutic paradox arises: autologous therapy using adult MSCs may be least effective in elderly patients—the very population most in need of treatment.
In contrast, perinatal MSCs such as WJ-MSCs do not carry these limitations. In murine models, WJ-MSCs promoted superior bone formation compared with BM-MSCs (62.5% vs. 25%), suggesting their potential role in sternal regeneration. In vivo studies have also demonstrated high survival rates and clear osteogenic superiority of WJ-MSCs. Moreover, WJ-MSCs or their secretome have proven effective in difficult-to-heal chronic wounds such as trophic ulcers; topical application of secretome/exosomes has been shown to shorten recovery time in diabetic ulcers. Perilesional infiltration of WJ-MSCs may modulate the local inflammatory response, reduce microbial burden, and promote adequate granulation tissue formation, thereby creating favorable conditions for chest wall reconstruction and restoration of sternal structure.
The combined advantages of WJ-MSCs—including non-invasive procurement, high proliferative capacity, independence from donor age, prominent osteogenic activity, and demonstrated efficacy in wound healing—formed the rationale for selecting WJ-MSCs in this clinical case. The results suggest that WJ-MSCs may serve as a potential adjunctive therapy to conventional corrective surgery, particularly in high-risk patients. The anticipated clinical goals include complete wound healing, chest wall stabilization, and absence of infection recurrence, even in elderly individuals.
However, this report has several limitations. First, independent or multicenter controlled studies evaluating WJ-MSCs in chronic sternal wounds are lacking; therefore, it cannot be conclusively stated that the observed effect was solely attributable to cell therapy. Second, the use of allogeneic cells inherently carries a potential risk of immune reactions. In this case, WJ-MSCs did not express HLA-DR (≤2% positive cells), consistent with baseline levels and indicating a low risk of immune activation. Nevertheless, in autoimmune or immunocompromised conditions, careful evaluation of immunological risk is warranted, particularly if systemic administration is considered. Accordingly, larger-scale studies are required to fully determine the long-term safety and adverse event profile of WJ-MSC therapy.
Conclusion
This case report suggests that allogeneic Wharton’s jelly-derived mesenchymal stem cells (WJ-MSCs) may promote sternal wound healing following coronary artery bypass grafting (CABG). In our case, the use of WJ-MSCs contributed to altering the course of refractory chronic sternal osteomyelitis in a high-risk elderly patient who was also COVID-19 positive.
The two-stage therapeutic strategy, consisting of thorough debridement of the infectious focus followed by WJ-MSC therapy, not only controlled and resolved the chronic inflammatory process but also significantly promoted regenerative processes at the site of injury. Our clinical experience indicates that WJ-MSCs hold therapeutic potential in complex cases of deep sternal wound infections.
The application of WJ-MSCs may be considered for broader use in clinical practice as a safe and effective therapeutic strategy. However, further prospective studies and larger case series are required to clearly define indications, optimize treatment protocols, and confirm long-term efficacy, thereby progressively integrating this approach into modern advanced surgical standards of care.
References
Ganina, A., Baigenzhin, A., Chuvakova, E., Yerzhigit, N., Zhunussov, A., Akhayeva, A., Kozina, L., Lookin, O., & Askarov, M. (2026). Intradermal Application of Allogenic Wharton’s Jelly Mesenchymal Stem Cells for Chronic Post-Thoracotomy Wound in an Elderly Patient After Coronary Artery Bypass Grafting: Clinical Case with Brief Literature Review. Diseases (Basel, Switzerland), 14(1), 27.
Source: Diseases