International Journal of Translational Medicine, 12/02/2026
Introduction
Chronic shoulder pain is one of the most common reasons for musculoskeletal consultation, ranking third in primary care after low back pain and knee pain. Common causes include rotator cuff disorders, adhesive capsulitis, calcific tendinitis, impingement syndrome, and degenerative joint changes. These conditions increase the healthcare burden, reduce work productivity, raise rates of sick leave, and may lead to early retirement or job loss.
Current treatment approaches mainly focus on pain control, physical therapy, lifestyle modification, and management of underlying causes, with surgical intervention required in many cases. However, a significant proportion of patients do not achieve the desired outcomes, continuing to experience pain and functional limitation. This highlights the need to develop tissue regenerative strategies rather than solely symptom control.
Mesenchymal stem cell (MSC)-based therapy has attracted considerable interest as a potential approach. MSCs are multipotent stromal cells capable of differentiating into bone, cartilage, muscle, and adipose tissue, and possess immunomodulatory, anti-inflammatory properties while secreting paracrine factors that promote tissue repair. Preclinical studies in shoulder injury models have shown that MSC therapy improves tendon–bone healing, enhances fibrocartilage formation, reduces inflammatory cell infiltration, and improves the biomechanical properties of the tissue.
Clinical data regarding the safety and efficacy of MSCs are increasingly expanding. Several studies have reported that this minimally invasive therapy is safe, reduces pain, and improves shoulder joint function following intra-articular injection. Intratendinous MSC injection for full-thickness rotator cuff tears has been reported to be safe over a 36-month follow-up period, with no local or systemic adverse events observed, while maintaining improvements in pain and shoulder function. However, unanswered questions remain due to heterogeneity in MSC manufacturing and application protocols.
The present study evaluates intra-articular injection of umbilical cord-derived mesenchymal stem cells (UC-MSCs) in patients with chronic shoulder pain, aiming to determine the safety and efficacy of this method as a regenerative alternative to conventional treatment strategies in a non-controlled setting.
Materials and Methods
Study Design, Location and Ethical Approval
This was a retrospective, observational before–after practice study based on medical records, conducted to evaluate changes in ASES scores in patients with chronic shoulder pain following intra-articular injection of a defined dose of human umbilical cord-derived mesenchymal stem cells (UC-MSCs).
The study was conducted at the Regenerative Medicine Institute (San José, Costa Rica) between June 2022 and April 2023. This is an outpatient clinic licensed by the Ministry of Health of Costa Rica to administer expanded allogeneic mesenchymal stem cell therapy.
The study protocol was approved by the ACIB-FUNIN Ethics Committee (January 16, 2024; code CEC-FUNIN-016-2023).
Population and Sample
A total of 20 medical records were selected using a non-probability convenience sampling method, based on the criteria of complete data and representativeness of the condition of interest. This sample size corresponds to all patients treated for shoulder pain at the Regenerative Medicine Institute during the period from June 2022 to April 2023.
The study included patients aged ≥18 years who were treated for chronic shoulder pain during the specified period. Exclusion criteria comprised: history of cancer (active or prior), medical records missing more than 50% of the required data, fewer than two recorded outcome assessment scores, or treatment received outside the study period.
Study Procedure and Intervention
Prior to the intervention, patients underwent ultrasound examination of the affected area. A trained physician performed a clinical assessment in combination with ultrasound imaging to determine the appropriate injection site.
Patients were then transferred to the procedure room and could receive mild sedation depending on personal preference. Human umbilical cord-derived mesenchymal stem cells (UC-MSCs) were administered at doses ranging from 7.8 to 38.6 million cells, adjusted according to clinical needs and individual ultrasound findings. The mean cell viability at the time of administration was 79.4%.
Collection and Processing of UC-MSCs
UC-MSCs were obtained from umbilical cords of full-term cesarean deliveries, following donor consent and maternal medical history screening. Maternal blood samples were tested serologically to exclude transmissible diseases; samples failing to meet safety criteria were discarded.
The umbilical cords were processed under sterile conditions, with blood vessels removed, minced, and tested for Mycoplasma spp., then cultured in alpha-MEM supplemented with 10% fetal bovine serum until reaching confluence and expanded to passage 5. Cells underwent quality control testing (bacteria, fungi, Mycoplasma), assessment of differentiation potential toward chondrogenic, osteogenic, and adipogenic lineages; surface marker analysis (positive for CD90, CD73, CD105, CD44; negative for CD34, CD19, CD45, CD11b); and karyotype evaluation.
Qualified cells were cryopreserved at −180°C. Prior to use, thawed cells were counted and viability was determined using trypan blue.
Data Collection
Data were extracted from electronic medical records using a standardized form, including demographic variables, medical history, anthropometric parameters, cell characteristics (cell count, viability), and post-treatment outcomes.
Efficacy was assessed using the ASES score; the minimal clinically important difference (MCID) was defined as 12 points.
Adverse events were recorded during follow-up and classified according to the Common Terminology Criteria for Adverse Events (CTCAE v4, 2017).
Statistical Analysis of Data
Descriptive analysis was performed and normality of the ASES change scores was assessed; the data met the assumption of normal distribution.
Pre–post 3-month ASES scores were compared using a paired t-test (with the Wilcoxon test applied for sensitivity analysis), and effect sizes were calculated (Cohen’s d, Hedges’ g).
An ANCOVA model and supplementary regression analyses were constructed to evaluate the association between UC-MSC dose and post-treatment functional outcomes, adjusted for baseline ASES score, age, and BMI. No longitudinal model was applied due to the availability of only one post-treatment assessment time point.
Results
Population Characteristics
A total of 20 patients received intra-articular UC-MSC injections during the period from June 2022 to April 2023. The primary treatment indication was chronic shoulder pain control (100% of cases).
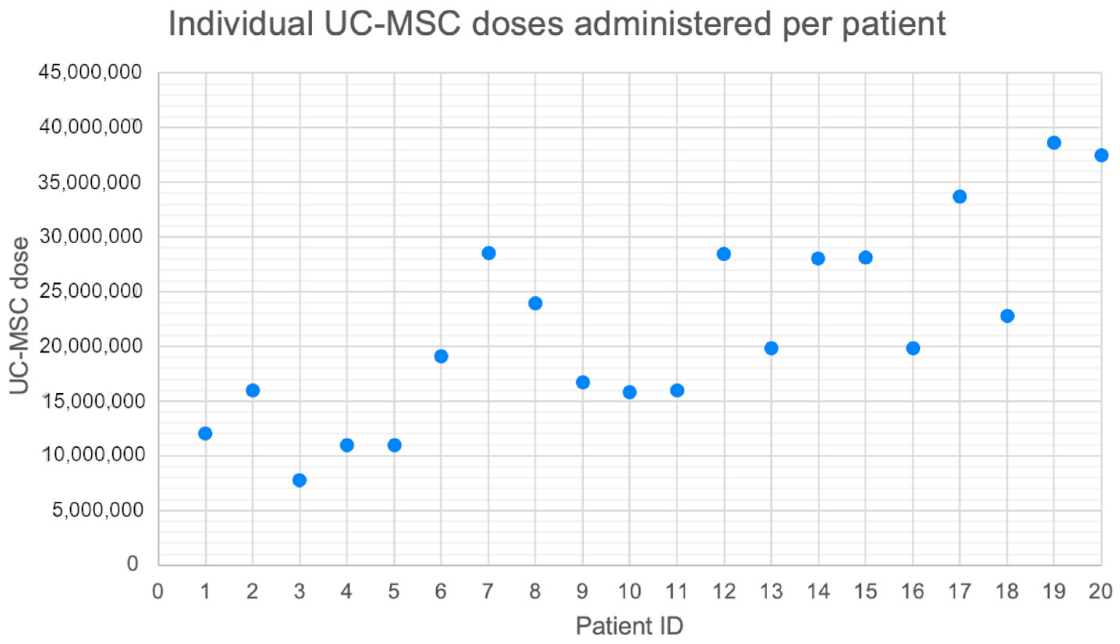
Figure 1. Individual UC-MSC doses administered per patient. Each point represents the total number of cells injected per joint for an individual participant, illustrating the variability associated with clinically individualized dosing.
Measurement of Treatment Outcome
Shoulder function improved significantly after treatment. The paired t-test showed that ASES scores at 3 months were significantly higher compared to baseline (p = 0.0004); the Wilcoxon test yielded consistent results (p = 0.008).
The mean improvement was 17 ASES points (Figure 2), reaching clinical significance. Effect size analysis demonstrated a large magnitude of effect, with Cohen’s d = 0.96 and Hedges’ g = 0.92 (adjusted for small sample size).
Regression Model
The ANCOVA model predicting ASES scores at 3 months explained approximately 26% of the variance and reached borderline statistical significance (p ≈ 0.06). In this model, only baseline ASES was a statistically significant predictor (p = 0.010), indicating that patients with better initial function tended to achieve higher follow-up scores.
The mean MSC dose per joint showed a positive correlation trend but did not reach statistical significance (p = 0.113).
The regression model using absolute ASES improvement as the dependent variable demonstrated a clearer association between MSC dose and clinical response. The model reached borderline significance (p = 0.0787) and explained 19% of the variance.
The MSC dose per joint variable achieved statistical significance (p = 0.039), indicating that higher doses were associated with greater improvements in shoulder function.
These findings suggest a potential dose–response relationship, more evident when directly analyzing the magnitude of improvement rather than post-treatment follow-up scores alone.
Adverse Events
During the 3-month follow-up period, no adverse events were recorded. No edema, erythema, bleeding, hematoma, infection, allergic reactions, bruising, vascular or nerve injury, hemarthrosis, or synovitis were observed during or immediately after the procedure.
At the 3-month assessment, no late complications commonly reported were identified, such as chronic pain, joint stiffness, osteomyelitis, septic arthritis, joint deformity, or vascular and nerve damage.
Discussion
This retrospective study showed that intra-articular UC-MSC injection significantly and clinically improved shoulder function in patients with chronic shoulder pain, with a mean increase of 17.17 ASES points after 3 months; 60% of patients exceeded the MCID threshold of 12 points.
These findings are consistent with previous studies on MSC therapy in shoulder disorders, including improvements in pain, range of motion, and patient-reported function. For example, Nejati reported sustained improvement over 36 months in patients with full-thickness rotator cuff tears treated with autologous adipose-derived MSCs; Vieira Ferreira also reported enhanced tendon healing and reduced re-tear rates when MSCs were used to augment surgery. However, differences in study design and outcome measures limit direct quantitative comparison.
Regression analysis indicated that baseline ASES was the main predictor of 3-month outcomes; patients with better pre-treatment function tended to maintain higher follow-up scores. The model analyzing absolute improvement suggested a dose–response relationship (higher MSC doses associated with greater improvement), although the small sample size warrants cautious interpretation. Age and BMI were not significantly associated with functional recovery.
The observed improvements are biologically plausible, based on preclinical evidence showing that MSCs modulate inflammation, promote extracellular matrix remodeling, and enhance tendon–bone integration. Clinically, UC-MSCs may represent a potential option for patients who do not respond to conservative treatment or who wish to avoid corticosteroids or surgery. No adverse events were recorded during the 3-month follow-up, further supporting the safety of the approach.
Major limitations include the retrospective uncontrolled design, small sample size, short follow-up duration, and lack of imaging assessment; therefore, structural regenerative effects cannot be confirmed. Randomized controlled trials with larger samples, standardized dosing, and long-term follow-up, incorporating MRI or ultrasound and biological markers, are needed to clarify mechanisms and optimize treatment protocols.
In conclusion, intra-articular UC-MSC injection is a safe and promising approach for the treatment of chronic shoulder pain, providing clinically meaningful short-term functional improvement; however, higher-quality evidence is required to confirm and standardize its application.
Conclusion
The study demonstrates that UC-MSC therapy has significant potential in improving function and reducing symptoms in patients with chronic shoulder pain, thereby contributing to enhanced quality of life and functional recovery.
Although limitations remain, the results achieved are meaningful both statistically and clinically, supporting consideration of this approach in a broader patient population. Future studies should confirm these findings in larger and more diverse samples, while also further evaluating the role of lifestyle factors in optimizing treatment outcomes.
References
Soto-Rodríguez, A., Deliyore-Vega, L. F., González-Kitzing, M., Muñoz-Araya, P. M., Valverde-Espinoza, J. A., Urzola-Herrera, V., Giampapa, V., & Rojas-Solano, J. R. (2026). Intra-Articular Application of Umbilical Cord-Derived Stem Cells in Patients with Chronic Shoulder Pain. International Journal of Translational Medicine, 6(1), 10
Source: International Journal of Translational Medicine