Frontiers in bioengineering and biotechnology, 25/09/2025
Human amniotic membrane-derived mesenchymal stem cells (hAMSCs) are considered promising candidates in regenerative medicine due to their multipotent differentiation capacity, tissue regeneration potential, low immunogenicity, and strong immunomodulatory properties. Compared to MSCs from bone marrow or adipose tissue, hAMSCs proliferate faster, have a lower risk of graft rejection, and are abundantly available, being obtained non-invasively and without ethical concerns. When combined with biomaterials such as natural polymers, synthetic polymers, or scaffolds, hAMSCs exhibit enhanced survival, proliferation, and integration into damaged tissues. Scaffolds designed with biocompatibility, biodegradability, and the ability to mimic the natural extracellular matrix (ECM) structure further enhance the regenerative capacity of these cells. This combined approach holds promise for developing more effective and personalized regenerative therapies for various chronic diseases and tissue degenerations.
Introduction
Regenerative medicine focuses on repairing and regenerating tissues and organs by replacing damaged cells. Mesenchymal stem cells (MSCs) serve as a key foundation, with the potential to restore various types of tissues and treat diverse pathological conditions. Among them, human amniotic membrane-derived mesenchymal stem cells (hAMSCs) stand out due to their unique biological properties, easily accessible source, and effective combination with biomaterials and scaffolds to promote tissue repair and regeneration. hAMSCs are isolated from the amniotic membrane of the human placenta—a thin, avascular, multilayered membrane that protects the fetus and secretes prostaglandins, particularly PGE₂, during labor.
Within the amniotic membrane, there are two main types of stem cells:
- i) Human amniotic epithelial cells (hAECs) – located in the innermost layer, directly in contact with the amniotic fluid, and derived from the embryonic ectoderm.
ii) Human amniotic membrane-derived mesenchymal stem cells (hAMSCs) – located within the stromal layer of the amniotic membrane and derived from the embryonic endoderm.
Human amniotic membrane-derived mesenchymal stem cells (hAMSCs) are isolated from the mesodermal layer of the amniotic membrane with high yield (~5 × 10⁸ cells per placenta). In vitro, hAMSCs are adherent, fibroblast-like cells that proliferate stably over 14 passages, with an average diameter of ~15 µm, and exhibit hybrid characteristics between epithelial and mesenchymal cells at the ultrastructural level. However, differences in isolation protocols across studies complicate comparison and interpretation of results; this review only analyzes studies that clearly specify the source of MSCs from the amniotic membrane.
Table 1. Key properties of hAMSCs.
| Property | Description |
| Origin | Derived from human amniotic membrane (mesodermal layer) |
| Morphology | Adherent, fibroblask-like; diameter 9 – 24 µm |
| Differentiation | Osteogenic, adipogenic, chondrogenic; also neuroglial, hepatic, pancreatic, cardiac, myogenic |
| Pluripotency Markers | OCT4, SSEA-3, SSEA-3 |
| Tumorigenicity | No evidence of tumor formation; low/no TERT expression |
| Immunogenicity | Low MHC-1, absent MHC-II; immune-privileged |
Table 2. Therapeutic applications of hAMSCs.
| Application area | Details |
| Bone/Cartilage Repair | Osteochondral defect repair, cartilage-like tissue regeneration |
| Fibrosis | Reduces liver, renal, and pulmonary fibrosis; modulates ECM genes |
| Cardiac/Vascular | Cardiomyocyte differentiation, agiogenesis, improved infacrct healing |
| Skeletal Muscle | VML repair, myogenic differentiation, Wnt signaling |
| Nervous System | Axonal regeneration, neuroprotection, inflammation reduction |
| Wound Healing | Promotes fibroblast migration, keratinocyte proliferation, anti-apoptotic effect |
Differentiation potential of hAMSCs
hAMSCs possess in vitro multipotent differentiation capacity, giving rise to osteogenic, adipogenic, and chondrogenic cells, as well as other cell types including neural, hepatic, pancreatic, cardiomyocyte, smooth muscle, and endothelial-like cells, demonstrating pronounced pluripotent potential. They express pluripotency markers such as OCT4, SSEA-3, and SSEA-4 but do not form tumors and exhibit very low or absent TERT activity. The differentiation capacity of hAMSCs is strongly influenced by culture conditions; repeated passaging in animal factor-free media can alter their phenotype and reduce their differentiation potential.
Angiogenic and antifibrotic activity of hAMSCs
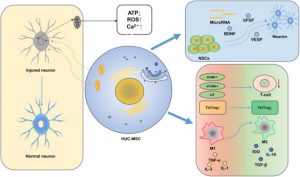
hAMSCs exhibit strong angiogenic and vasculogenic potential, with high expression of angiogenic factors such as VEGF-A, angiopoietin-1, HGF, FGF-2, and the anti-apoptotic factor AKT-1. In vitro, hAMSCs undergo morphological changes during endothelial differentiation but show limited expression of mature endothelial markers, suggesting a self-regulatory mechanism and primarily indirect angiogenic effects. Additionally, hAMSCs can inhibit angiogenesis and reduce inflammation, as demonstrated in an alkali-induced corneal injury model, where they decreased neovascularization, corneal opacity, and inflammatory cell infiltration.
Proliferative and anti-apoptotic effects of hAMSCs
Human amniotic membrane-derived mesenchymal stem cells (hAMSCs) contribute to wound healing and tissue regeneration through their secreted factors (secretome) by activating PI3K/Akt signaling pathways, promoting epidermal cell proliferation, inhibiting apoptosis, and enhancing fibroblast migration.
Additionally, hAMSCs have been applied in the construction of 3D skin structures. In oncology and reproductive medicine, cytokines secreted by hAMSCs, such as FGF-2, IGF-1, HGF, VEGF, and EGF, have demonstrated proliferative and anti-apoptotic effects in models of ovarian cancer and premature ovarian failure.
Immunomodulation and immunosuppression of hAMSCs
hAMSCs exhibit remarkable immunomodulatory properties and high therapeutic potential:
- hAMSCs possess an immune-privileged status, playing a key role in maintaining maternal-fetal immune tolerance. They express low levels of MHC-I, lack MHC-II and co-stimulatory molecules (CD40, CD80, CD86), helping them evade immune recognition.
- No acute graft rejection has been observed following hAMSC or hAEC transplantation in humans or animals; these cells can persist for at least 2 months in grafted tissue.
- hAMSCs influence multiple immune cell types (T cells, B cells, NK cells, dendritic cells, macrophages, neutrophils) through both cell–cell interactions and soluble factors such as IL-6, PGE2, TGF-β, NO, IDO, HGF, IGF-1.
- Host inflammatory cytokines can activate hAMSCs to secrete immunoregulatory factors and promote tissue repair, creating a complex, context-dependent bidirectional immunoregulatory network.
- hAMSCs inhibit monocyte differentiation into dendritic cells, induce M2 macrophages, and suppress T cell proliferation, demonstrating pronounced anti-inflammatory effects.
- These effects are dose-dependent and require direct cell contact in combination with soluble factors (PGE2, IL-10, TGF-β, NO, IDO).
In summary, hAMSCs not only share the characteristics of other MSCs (BM-MSCs, ADSCs) but also express additional pluripotency markers, are non-tumorigenic, easily obtainable, ethically uncontroversial, and stable across age and culture conditions, making them a highly promising MSC source for cell therapy and regenerative medicine. Recent preclinical studies have also demonstrated the tissue regenerative efficacy of hAMSCs across various disease models.
Bone and cartilage injuries
Human amniotic membrane-derived mesenchymal stem cells (hAMSCs) have been applied in the treatment of bone and cartilage injuries, revealing potential for clinical bone–cartilage regeneration. hAMSC sheets encapsulating cartilage particles have shown efficacy in repairing bone–cartilage defects in rabbits, promoting the formation of hyaline-like cartilage tissue with better integration into surrounding healthy cartilage and superior subchondral bone regeneration compared to control groups. The study by Muiños-López’s team also demonstrated that combining hAMSCs with human amniotic membrane (hAM) enhances cartilage repair to a level comparable to mature chondrocytes. Furthermore, in micromass culture models, hAMSCs are capable of differentiating into chondrocytes, confirming their clinical application potential for cartilage injury treatment.
Fibrosis
Human amniotic membrane-derived mesenchymal stem cells (hAMSCs) demonstrate significant efficacy in treating tissue fibrosis, particularly liver fibrosis. They inhibit the activation of hepatic stellate cells (HSCs), reduce collagen deposition, and suppress Wnt signaling through secreted factors such as IGFBP-3, DKK-1, and DKK-3. In addition, hAMSCs upregulate the expression of extracellular matrix-degrading genes (MMP-2, MMP-9, MMP-13, TIMP-1), contributing to the attenuation of fibrosis in the liver, kidney, and lungs.
Through their secretome and exosome-rich conditioned media (ERCM), hAMSCs also promote skin wound healing by stimulating epidermal cell proliferation, activating fibroblasts, inducing angiogenesis, and modulating scar tissue formation.
Cardiac pathologies
In preclinical models of ischemic heart disease, human amniotic membrane-derived mesenchymal stem cells (hAMSCs) have demonstrated the ability to restore cardiac function and reduce infarct size. Transplanted cells can differentiate into cardiomyocyte-like cells, while hAMSC-conditioned media also provide cardioprotective effects, promote angiogenesis, and attenuate myocardial fibrosis.
Vascular pathologies
Preclinical studies have shown that human amniotic membrane-derived mesenchymal stem cells (hAMSCs) possess the ability to regenerate muscle and vasculature in various injury models:
- In volumetric muscle loss (VML) models, hAMSCs induced to undergo myogenic differentiation with 5-azacytidine exhibited enhanced angiogenesis and muscle tissue restoration, mediated by activation of the Wnt/β-catenin signaling pathway.
- When implanted into injured bladder walls, hAMSCs differentiated into smooth muscle cells, promoting regeneration of the muscular layer.
- Direct transplantation of hAMSCs into ischemic limbs increased perfusion, capillary density, and subcutaneous blood flow, suggesting that their pro-angiogenic effects are primarily mediated by soluble factors secreted by the cells.
Central nervous system
Human amniotic membrane-derived mesenchymal stem cells (hAMSCs) have demonstrated significant efficacy in central nervous system (CNS) recovery, including neuroprotection, neuronal regeneration, and improvement of motor function.
- In acute spinal cord injury models in mice, intravenous administration of hAMSCs improved sensory function and gait, reduced inflammation, decreased ED1-positive inflammatory cells and cytokines (TNF-α, IL-6, IL-1β), while also reducing apoptosis, enhancing angiogenesis, and promoting axonal regeneration.
- hAMSCs also stimulate axonal regeneration in injured retinal neurons in mice, restoring neural activity under both normal and hypoxic conditions.
Although the therapeutic potential is considerable, clinical application of hAMSCs in neurology remains at an early stage. However, combining hAMSCs with scaffolds and biomaterials is opening new avenues in tissue engineering and neural functional recovery.
Biomaterials, scaffolds and hAMSCs
Biomaterials are materials that interact favorably with biological systems and can be either naturally derived or artificially designed to replace, restore, or enhance the function of tissues and organs. In regenerative medicine, the use of biomaterials has developed rapidly, particularly in tissue engineering and orthopedic applications.
When combined with mesenchymal stem cells (MSCs), including human amniotic membrane-derived mesenchymal stem cells (hAMSCs), biomaterials are considered one of the most promising approaches for treating various diseases. This combination helps to:
- Optimize the biological microenvironment necessary for cell regeneration
- Enhance therapeutic efficacy and tissue functional recovery
- Improve integration between the implant/scaffold and the patient’s tissue
Biomaterials can be classified based on their origin and properties into metals, polymers, ceramics, composite materials (combining polymers with ceramic or metal particles), and natural biomaterials.
Table 3. Types of biomaterials and scaffolds used with hAMSCs.
| Biomaterial type | Examples | Application |
| Metallic | Titanium, Au-Ni nanowires | Neural interface, implants |
| Polymeric | Hydrogels, GelMA, PPCNg, PLLA/PEG | 3D culture, injectable therapies |
| Ceramics | Hydroxyapatite, Bio-Oss | Bone regeneration |
| Natural | Collagen, fibrin, chitosan | Soft tissue repair, wound healing |
| Hybrid/ECM | Decellularized amnion, ECM from hAMSCs | Wound healing, CNS repair |
Table 4. Current strategies and advances in combining hAMSCs with biomaterials for regenerative medicine applications.
| Biomaterial and scaffold | Outcomes/improvements |
| Combination of metallic, semiconductor or oxide nanomaterials in polymers | Higher osteo and chondrogenic differentiation |
| Hydrogels | – Retention/preservation hAMSC phenotype, stem marker expression, and differentiation capabilities
– Significant/marked increase in the synthesis and secretion of factors |
| Hydrogel composed of GelMA | – Increases the number of mature neurons to repair TBI damage
– (higher expression) in the levels of NeuN, NSF, BDNF, and MAP2 |
| RGDmix hydrogel incorporationg the RGDSP peptide | Increases hAMSC survival at the transplantation site |
| Biodegradable poly (L-lactide) (PLLA) | Improved urethral defect repair in rabbit animal models (including morphology, tissue reconstruction, and complication incidence) and enhanced re-epithelialization |
| Mineral matrices | Significantly increasing new bone formation (paracine effect) |
| Chitosan, hyaluronic acid and carbonate apatite hydrogels | – Enhance hAMSC proliferation, promote neovascularization and reduce fibrosis
– Higher proliferation and osteo differentiation |
| Silk fiber | Promotion osteo differentiation into the in vitro and in vivo, the secretion of angiogenesis related factors |
Human amniotic membrane-derived mesenchymal stem cells (hAMSCs) are highly valued for their therapeutic potential in chronic inflammatory diseases, musculoskeletal disorders, and neurological conditions. However, clinical success depends on maintaining cell functionality and viability after transplantation. Combining hAMSCs with biomaterials and scaffolds provides mechanical support and structural cues to guide tissue regeneration while protecting cells from adverse microenvironments. Natural or synthetic scaffolds, particularly when functionalized with growth factors or extracellular matrix (ECM) components, have been shown to enhance hAMSC proliferation, differentiation, and paracrine activity in preclinical models.
Numerous in vivo studies indicate that integrating hAMSCs with advanced biomaterials significantly improves skin wound healing, corrects bone defects, and restores central nervous system injuries by enhancing cell survival, promoting angiogenesis, and accelerating functional recovery. Despite these promising preclinical results, clinical application remains limited due to donor variability, challenges in scaling up cell production under GMP standards, and a lack of standardized protocols. In the future, developing tissue-specific scaffolds alongside improvements in production and cell delivery technologies will be key to fully harnessing the therapeutic potential of hAMSCs and advancing them toward clinical use.
References
Source: Elvira H. de Laorden, Beatriz L. Rodilla, María Arroyo-Hernández, Maite Iglesias (2025), Advances in human amniotic placenta membrane-derived mesenchymal stromal cells (hAMSCs) for regenerative medicine: enhancing therapeutic potential with biomaterials and scaffolds, Frontiers in bioengineering and biotechnology, Volume 13.
Link: https://www.frontiersin.org/journals/bioengineering-and-biotechnology/articles/10.3389/fbioe.2025.1590393/full?utm_source=chatgpt.com#B114