Journal of Neurorestoratology, 12/02/2026
Introduction
Cerebral palsy (CP) is a group of non-progressive disorders of the central nervous system caused by brain injury during the fetal or neonatal period, characterized by motor and postural dysfunction. Numerous risk factors have been identified, such as perinatal brain injury, intrauterine infection, prematurity, congenital malformations, and abnormal fetal inflammatory responses. CP not only affects motor function but also causes language, cognitive, and psychological impairments, creating a significant economic and social burden.
Currently, treatment mainly includes rehabilitation, antispastic medications, and surgical interventions. However, these approaches only improve symptoms and cannot restore the underlying neurological damage, resulting in limited long-term efficacy. Therefore, there is an urgent need for novel therapies capable of neural regeneration and functional recovery. In this context, cell therapy has emerged as a promising approach in the treatment of neurological disorders, including cerebral palsy.
Cell transplantation therapy as an emerging regenerative medicine approach
Cell transplantation therapy is an emerging approach in regenerative medicine, demonstrating significant potential in the treatment of various diseases. Stem cells possess the abilities of proliferation, self-renewal, and differentiation, with the main types used including HSCs, ESCs, iPSCs, NSCs, and MSCs. Among these, mesenchymal stromal cells (MSCs) have self-renewal and multilineage differentiation capabilities and have been applied in numerous clinical trials. However, MSCs derived from bone marrow or adipose tissue still have limitations, such as reduced potential with age or invasive collection procedures. In contrast, mesenchymal stromal cells from umbilical cord tissue (UC-MSCs) exhibit several notable advantages: abundant source, non-invasive collection, minimal ethical concerns, low immunogenicity, and high safety, making them suitable for clinical applications. Recently, UC-MSCs have demonstrated significant efficacy in neural regeneration and recovery through mechanisms including the secretion of neurotrophic factors, immunomodulation, and anti-inflammatory effects, opening a promising therapeutic avenue for cerebral palsy.
Methodology
This study employed a literature review approach. A total of 32 articles were selected to synthesize descriptions of study design, sample characteristics, intervention methods, and main outcomes.
Preclinical research on hUC-MSCs
Mechanisms of hUC-MSCs action in neurorestoration
Hypoxia–ischemia causes brain injury in cerebral palsy by inducing oligodendrocyte death, myelin loss, and activation of glial cells, leading to oxidative stress and inflammatory responses with cytokines such as IL-1, IL-6, TNF-α, and IFN-γ. The consequence is prolonged neuroinflammation, blood–brain barrier disruption, inhibition of axonal regeneration, and secondary injury.
Therefore, treatment needs to focus on myelin regeneration, inflammation modulation, and neural network restoration through mechanisms such as regeneration, protection, and enhancement of neural plasticity—this forms the basis for the application of hUC-MSCs.
(Ⅰ) Immunomodulation and Microenvironment Remodeling
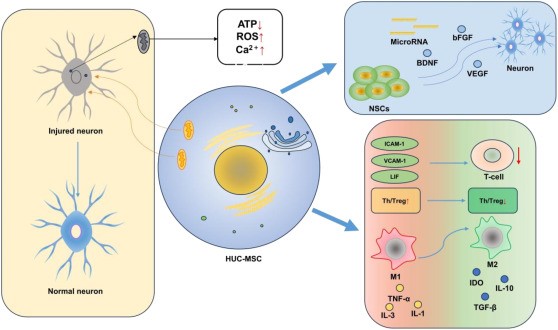
Fig. 1. In the central nervous system, hUC-MSCs promote neural repair through three main mechanisms: immunomodulation (red–green), paracrine signaling (blue), and organelle transfer (yellow).
hUC-MSCs modulate the immune system by suppressing T cell responses, reducing antigen presentation, and restoring Th17/Treg balance. At the site of injury, they promote M1-to-M2 polarization, decreasing pro-inflammatory cytokines (TNF-α, IL-1β) and increasing anti-inflammatory factors (IL-10), thereby reducing neuroinflammation and supporting tissue regeneration.
Additionally, hUC-MSCs secrete LIF, TGF-β, IL-10, and IDO, which help inhibit oxidative stress and glial cell activation, creating a favorable immunometabolic microenvironment for neural recovery through a “first immunomodulation, then repair” mechanism.
(Ⅱ) Neurotrophic Support and Neural Regeneration
hUC-MSCs exert their effects through a paracrine mechanism, secreting neurotrophic factors such as BDNF, VEGF, and bFGF, which enhance neuronal survival, support recovery, and promote neurogenesis. At the same time, hUC-MSCs activate endogenous neural stem cells (NSCs) and guide their differentiation into functional neurons.
Additionally, hUC-MSCs stimulate the expression of microRNAs (miR-124, miR-145), contributing to the proliferation and differentiation of neural progenitor cells, thereby promoting neural regeneration.
(Ⅲ) Proliferation, Replacement, and Mitochondrial Rescue
Beyond paracrine mechanisms, hUC-MSCs directly contribute to neural repair through:
- Neural differentiation: Within the injured microenvironment, hUC-MSCs can proliferate and differentiate into neuron-like cells, contributing to the replacement of damaged cells and the restoration of neural function.
- Mitochondrial transfer (MT): Mitochondrial dysfunction (reduced OXPHOS, ATP depletion, increased oxidative stress, and disrupted Ca²⁺ homeostasis) exacerbates neural injury. hUC-MSCs serve as a source of mitochondria, establishing organelle exchange with recipient cells, restoring ATP production (~45%), and enhancing the proliferation capacity of recipient cells.
This mechanism highlights the role of mitochondria as a critical “functional factor” in cellular repair and opens the avenue for organelle-based therapies in cerebral palsy treatment.
Animal studies in CP treatment
Animal studies are a crucial step for evaluating the efficacy, safety, and mechanisms of hUC-MSCs before clinical translation. Rats and mice are commonly used models, capable of recapitulating motor, cognitive deficits, and brain lesions similar to cerebral palsy.
Results show that, at doses of 5 × 10³ – 5 × 10⁶ cells, hUC-MSCs improve motor and cognitive function while reducing brain lesion volume as assessed by MRI and histology. At the cellular level, this therapy decreases glial cell and astrocyte activation, inhibits oxidative stress (PKA/PKC), reduces autophagy and apoptosis (Beclin-2, caspase-3), and lowers proinflammatory cytokines (TNF-α, IL-1β).
Therapeutic efficacy depends on dose, frequency, timing, and administration route: higher doses and repeated injections yield better outcomes; early transplantation (within 24 hours post-injury) is more effective than late; and intravenous delivery facilitates greater migration to the brain compared with other routes.
Limitations of translating animal models to clinical practice
Although hUC-MSCs demonstrate efficacy in animal models, clinical translation remains limited due to physiological and brain developmental differences between rodents and humans, as well as the more complex brain structure in humans. Current models do not fully recapitulate all cerebral palsy features (spasticity, dystonia, cognitive deficits), and behavioral assessments remain limited. Additionally, standardizing cell doses is challenging due to interspecies body size differences.
→ Higher-order animal models and optimization of dose, timing, and administration route are needed to improve clinical applicability.
Clinical research on hUC-MSCs
Although clinical data on hUC-MSCs for cerebral palsy are still limited compared to preclinical studies, recent research indicates considerable potential of cell therapy. These studies provide preliminary evidence on the efficacy and safety of hUC-MSCs, while focusing on key factors such as cell dose, route of administration, timing of treatment (patient age), and study limitations.
Cell Dosage
Cell dose is an important factor determining therapeutic efficacy. Current studies suggest a dose range of 1 × 10⁶ – 1 × 10⁷ cells/kg as both safe and effective. A retrospective study of 152 children with cerebral palsy showed that a dose of 1 × 10⁶ cells/kg was well tolerated, with no serious adverse events, and contributed to improvements in motor function and muscle strength.
However, the optimal dose also depends on the administration route: intravenous (IV) infusion generally favors lower doses or concentrations to reduce infusion reactions, whereas intrathecal (IT) administration may allow higher doses or concentrations with potentially greater efficacy.
Cell transplantation route
Common routes for delivering hUC-MSCs include IV (intravenous), IT (intrathecal), IA (intra-arterial), IN (intranasal), and ICV (intracerebroventricular), but no optimal route has been definitively established.
- ICV: Direct injection into the brain → precise and rapid delivery; however, highly invasive, with risks of tissue damage and infection, making it clinically unsuitable, especially in children.
- IA: Less invasive than ICV, utilizes the “first-pass effect” through the brain to increase cell distribution; however, carries risks of embolism, ischemia, and cardiopulmonary complications.
- IV: The most commonly used route (~80%), safe and easy to perform; however, cells are prone to sequestration in the liver, lungs, and spleen, reducing the number reaching the brain.
- IT: Injection into cerebrospinal fluid → direct access to the central nervous system; requires high technical skill, strict asepsis, may require anesthesia, and carries risk of complications.
- IN: Non-invasive, can bypass the blood–brain barrier; however, absorption is limited by enzymatic degradation, mucosal pH, and individual variability, reducing efficacy.
→ Each route has specific advantages and limitations, and selection should be optimized based on treatment goals and patient condition.
Timing of cell transplantation (patient age)
The timing of cell transplantation is a critical factor affecting therapeutic efficacy. Current studies have primarily been conducted in pediatric patients (from neonates to approximately 16 years old), with younger patients generally showing better responses.
The pre-school period is considered a “golden window” because the nervous system is undergoing rapid development, enhancing the potential for neural repair and reorganization. A recent retrospective study also indicated that younger patients responded better to treatment, underscoring the importance of early intervention.
Safety
hUC-MSCs have been evaluated as relatively safe in clinical studies, with no serious adverse events reported. Common side effects include transient fever, headache, nausea, and back pain, most of which resolve spontaneously or improve within 72 hours with symptomatic treatment. Randomized controlled clinical trials have also confirmed this safety profile, showing only mild adverse events and no new complications during long-term follow-up (12–24 months).
Efficacy
hUC-MSCs have demonstrated significant improvement in gross motor function in patients with cerebral palsy. Efficacy has been evaluated using various scales and tools, including GMFM, FMFM, ADL, quality of life assessments, MRI/DTI, EEG, and brain network analysis. Clinical studies have reported significant increases in GMFM and FMFM scores, as well as muscle strength after treatment. The therapeutic effect tends to improve with the number of treatment sessions, with patients receiving multiple courses showing greater functional gains.
In addition, hUC-MSCs help reduce muscle tone (spasticity), particularly when combined with rehabilitation therapy, thereby enhancing overall motor function.
Analysis of key factors influencing therapeutic efficacy
Association between patient characteristics and treatment outcomes
(I) Impact of Sample Size and Age on Motor Function
FMFM outcomes depend on both sample size and patient age:
- Large sample + younger patients → greater improvement (due to higher neural plasticity).
- Small sample + older patients → differences are harder to detect, due to reduced responsiveness and limited statistical power.
→ Future studies should increase sample size and stratify patients by age more clearly.
(II) The Role of Sample Size in Cognitive Function Improvement Disparities
Small sample sizes increase the influence of individual variability, resulting in inconsistent outcomes across studies. Therefore, future studies should:
- Increase sample size and adopt multicenter, double-blind, controlled designs.
- Stratify patients by age in detail to reduce error.
- Standardize dosing by body weight (cells/kg) to improve precision and clinical applicability.
Limitations in current efficacy assessment systems
- Deficiencies in Assessment Tools
- Inadequate Follow-up Duration
Summary and outlook
hUC-MSCs show significant potential for neural repair in cerebral palsy through immunomodulation, inflammation reduction, and neuronal cell restoration, with preliminary evidence indicating short-term safety and efficacy. However, large-scale, multicenter, and long-term studies are still needed to confirm these findings. Current major challenges include limited cell sources, poor targeting accuracy, low utilization efficiency, and difficulty tracking cells in vivo.
Future directions include:
- Large-scale cell production, standardized protocols, and establishment of cell banks.
- Improving precision in cell delivery.
- Enhancing in vivo cell tracking (e.g., MRI with specialized labeling agents).
- Optimizing outcome assessment systems by integrating MRI, EEG, and AI to analyze brain function and predict treatment responses.
References
Sai Wang, Yachen Wang, Jing Liu (2026). Research progress on the therapeutic efficacy of human umbilical cord mesenchymal stromal cells for cerebral palsy in children. Journal of Neurorestoratology, Volume 14, Issue 2.
Source: Journal of Neurorestoratology
Link: https://www.sciencedirect.com/science/article/pii/S2324242625000889