Health Science Reports, 08/02/2026
Introduction
Pediatric kidney diseases encompass a wide range of disorders, including chronic kidney disease (CKD), acute kidney injury (AKI), glomerulonephritis, nephrotic syndrome, and congenital anomalies of the kidney and urinary tract (CAKUT). These conditions can significantly affect the growth, development, and overall health of children, often leading to long-term complications and reduced quality of life. Current treatment strategies mainly focus on symptom management and slowing disease progression; however, they often fail to address the underlying causes and do not effectively promote tissue regeneration.
In recent years, stem cell therapy has attracted considerable attention as a potentially transformative approach for the treatment of kidney diseases. Among various stem cell types, mesenchymal stem cells (MSCs) have emerged as particularly promising candidates for therapeutic applications in pediatric nephrology. MSCs are multipotent stem cells that can be isolated from several sources, including bone marrow, adipose tissue, umbilical cord, and dental pulp. These cells possess several properties favorable for regenerative medicine, such as self-renewal capacity, the ability to differentiate into multiple cell lineages, and immunomodulatory functions.
This review focuses on current research regarding the potential application of MSCs in the treatment of pediatric kidney diseases. Specifically, we summarize both preclinical and clinical studies investigating MSC-based therapies for various renal disorders, analyze their mechanisms of action, and discuss the potential benefits as well as the challenges associated with this emerging therapeutic strategy.
Mesenchymal Stem Cells: Characteristics and Sources
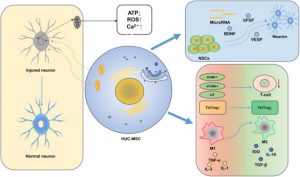
- Mesenchymal stem cells (MSCs) are multipotent stem cells capable of self-renewal and differentiation into bone, cartilage, and adipose tissue. According to the criteria of the International Society for Cell & Gene Therapy (ISCT), MSCs must adhere to plastic under culture conditions, express the surface markers CD73, CD90, and CD105, and lack expression of hematopoietic markers such as CD45, CD34, and HLA-DR.
- MSCs have therapeutic potential in kidney diseases due to their immunomodulatory and anti-inflammatory properties, including the ability to suppress T-cell activity and influence B cells and dendritic cells. In addition, they secrete growth factors and cytokines that enhance cell survival, proliferation, and angiogenesis. MSCs also exhibit low immunogenicity, which reduces the likelihood of immune rejection.
- Various MSC sources can be used for the treatment of pediatric kidney diseases, each with its own advantages and limitations. Bone marrow–derived MSCs (BM-MSCs) have been extensively studied and have shown promising results in kidney disease treatment. However, bone marrow aspiration is an invasive procedure, which may limit its application in pediatric patients. Adipose tissue–derived MSCs (AD-MSCs) represent a more accessible cell source that can be obtained through less invasive procedures and have demonstrated therapeutic efficacy comparable to BM-MSCs in some studies. Umbilical cord–derived MSCs (UC-MSCs) offer several advantages, including non-invasive collection, high proliferative capacity, and potentially superior immunomodulatory properties compared with MSCs derived from adult tissues.
- When selecting an MSC source for the treatment of pediatric kidney diseases, factors such as ease of collection, proliferative potential, immunomodulatory properties, and tissue regenerative capacity should be considered; therapeutic efficacy may vary depending on the cell source and the disease model.
Mechanisms of Action of MSCs in Kidney Diseases
- Immunomodulation: MSCs suppress the activation and proliferation of T cells, promote regulatory T cells (Tregs), inhibit B cells and dendritic cells, and induce macrophage polarization from the pro-inflammatory M1 phenotype to the anti-inflammatory M2 phenotype.
- Support of Tissue Regeneration: MSCs secrete growth factors that enhance the survival and proliferation of renal cells, promote angiogenesis, and may transfer healthy mitochondria to injured cells.
- Anti-fibrotic Effects: MSCs inhibit the epithelial-to-mesenchymal transition (EMT), reduce fibroblast formation, and promote the degradation of extracellular matrix components.
- Paracrine Effects: MSCs secrete numerous growth factors and anti-inflammatory cytokines (HGF, IGF-1, EGF, IL-10, TGF-β) and release extracellular vesicles/exosomes, which facilitate intercellular signaling and promote tissue repair.
Delivery Methods of MSC Therapy in Pediatric Kidney Disorders
- Intravenous (IV) injection: This is the most common and least invasive method, making it suitable for pediatric patients. MSCs migrate toward injured tissues via the SDF-1/CXCR4 axis. However, renal delivery efficiency is low due to the pulmonary first-pass effect, with only about 2–5% of cells reaching the kidneys.
- Systemic administration: This approach primarily exploits the paracrine effects of MSCs, such as exosomes, to modulate immune responses and slow disease progression (e.g., chronic kidney disease and nephrotic syndrome). Repeated administration is possible, but it faces limitations similar to IV delivery, including low renal homing and the potential risk of immune responses when using allogeneic MSCs.
- Local infusion: MSCs can be administered via the renal artery or directly into the renal parenchyma/subcapsular space, which increases the proportion of MSCs reaching the kidneys (approximately 10–20%). This approach may enhance renal tissue repair and reduce proteinuria. However, it is more invasive, requires interventional procedures, and carries risks such as bleeding and infection.
- Direct kidney infusion: MSCs are injected directly into the kidney parenchyma, allowing a high local concentration of cells at the injury site and showing effectiveness in glomerular injury. However, this approach is highly invasive, associated with higher procedural risks, and has limited impact on systemic inflammation.
The choice of MSC delivery method should balance therapeutic efficacy, invasiveness, and safety in pediatric patients. Future research should focus on improving MSC homing to the kidney and developing optimized delivery strategies for pediatric renal diseases.
Preclinical Studies on MSC Therapy for Pediatric Kidney Disorders
Preclinical studies (in animal models) have demonstrated that MSCs can reduce kidney injury, decrease inflammation, and promote tissue regeneration in pediatric kidney diseases.
- Chronic Kidney Disease (CKD): MSCs help slow disease progression, reduce renal fibrosis, decrease extracellular matrix (ECM) deposition, and modulate immune responses.
- Acute Kidney Injury (AKI):
+ In an ischemia–reperfusion injury (IRI) mouse model, BM-MSCs (1–2 × 10⁶ cells, intravenous injection) reduced serum creatinine levels by 30–50% and decreased inflammatory cytokines (TNF-α, IL-6).
+ UC-MSCs in a cisplatin-induced AKI model reduced apoptosis, decreased oxidative stress, and improved glomerular filtration rate (GFR).
- CKD in the 5/6 nephrectomy model: AD-MSCs (2 × 10⁶ cells, intraperitoneal injection) reduced interstitial fibrosis, decreased proteinuria (~40%), and lowered blood urea nitrogen (BUN) levels.
- Glomerular disorders: UC-MSCs (1 × 10⁶ cells, intravenous injection) in an anti-Thy1 glomerulonephritis model reduced proteinuria, decreased glomerular inflammation, and protected podocytes.
- Congenital kidney anomalies (UUO model): Studies in adult animal models suggest that MSCs can reduce fibrosis and preserve nephron formation; however, limited studies have been conducted in neonatal models.
Various animal models (IRI, nephrectomy, UUO), different MSC sources (BM-MSCs, UC-MSCs, AD-MSCs), and multiple evaluation parameters (creatinine, GFR, proteinuria, histology) have provided a strong preclinical foundation for the development of MSC therapy in pediatric kidney diseases.
Clinical Studies on MSC Therapy for Pediatric Kidney Disorders
Early clinical studies suggest that MSC therapy has potential safety and efficacy in the treatment of pediatric kidney diseases, including AKI, CKD, glomerular diseases, and CAKUT.
- Acute Kidney Injury (AKI):
+ A phase I trial in children aged 2–12 years (n = 15) used allogeneic BM-MSCs administered intravenously (1 × 10⁶ cells/kg).
+ Serum creatinine decreased by approximately 25% after 7 days, the need for dialysis decreased from 40% to 20%, and no serious adverse events were reported.
- Chronic Kidney Disease (CKD):
+ A phase I study in children aged 5–15 years (n = 12) used UC-MSCs (2 × 10⁶ cells/kg).
+ Estimated glomerular filtration rate (eGFR) remained stable over 6 months, while the albumin-to-creatinine ratio (ACR) decreased by approximately 30%. Mild adverse effects were observed (low-grade fever in ~10% of patients).
- Steroid-Resistant Nephrotic Syndrome (SRNS):
+ Children aged 3–10 years (n = 10) received autologous AD-MSCs (1 × 10⁶ cells/kg).
+ Proteinuria decreased by approximately 40% after 3 months, serum albumin increased, and edema improved in 60% of patients.
- Congenital Anomalies of the Kidney and Urinary Tract (CAKUT):
+ Studies related to the Wnt/β-catenin signaling pathway suggest that MSCs may improve renal structure.
+ An increase in renal cortical thickness of approximately 20% and a reduction in hydronephrosis severity were observed within 6 months.
Initial clinical data indicate that MSCs may improve kidney function and stabilize disease progression with relatively high safety; however, larger studies and long-term follow-up are needed to confirm their efficacy.
In addition to these early clinical findings, advances reported during 2023–2024 have further strengthened and expanded our understanding of the potential of MSC therapy in pediatric kidney diseases. MSCs are considered highly promising due to their regenerative and immunomodulatory properties, particularly given that the global mortality rate associated with acute kidney injury (AKI) remains high (20–50%). These cells can reduce renal inflammation by inhibiting the maturation of dendritic cells, thereby limiting inflammatory responses associated with disease progression. Recently, a novel approach has emerged involving the use of MSC-derived extracellular vesicles (MSC-EVs), which are approximately 30–100 nm in size and exhibit anti-inflammatory, anti-senescence, and tissue repair–promoting effects. Due to these properties, MSC-EVs are considered a promising “cell-free” therapeutic strategy, offering advantages in stability and safety compared with direct cell-based therapies, particularly for the treatment of pediatric kidney diseases.
MSC Therapy in Adult Kidney Diseases and Comparison With Pediatric Applications
- In adults, MSC therapy has been widely investigated in various kidney diseases, including AKI, CKD, glomerular diseases, post-transplant kidney disease, and diabetic nephropathy. MSCs can reduce inflammation, promote tubular repair, decrease fibrosis, and improve renal function, primarily through paracrine mechanisms.
- The NEPHSTROM trial (2023) demonstrated that MSC therapy could slow the decline in estimated glomerular filtration rate (eGFR) over 18 months, highlighting its therapeutic potential for kidney disease and providing a reference basis for pediatric research.
- Evidence reported in 2023 confirmed that MSCs exert anti-apoptotic, antioxidant, anti-inflammatory, and anti-fibrotic effects, partly mediated by extracellular vesicles (exosomes/EVs). MSCs and EVs also show therapeutic potential in virus-induced AKI.
- Recent advances specific to pediatrics (2024):
+ Extracellular vesicles derived from autologous renal cells (RACev) can reduce fibrosis, inflammation, and hypoxia following AKI while promoting angiogenesis.
+ The REACT trials apply an allometric scaling dosing strategy based on biological size correlation, using kidney mass estimated from 3D imaging (3 × 10⁶ cells/g), to personalize therapeutic dosing for pediatric patients.
Key Differences Between MSC Applications in Adults and Children
- Disease etiology:
- Adults: Mainly caused by diabetes, hypertension, and ischemia.
- Children: More commonly associated with congenital anomalies (CAKUT) or genetic disorders.
- Therapeutic goals:
- Adults: To slow down or reverse kidney damage.
- Children: To support kidney development and prevent early disease progression.
- Physiological characteristics and treatment approaches:
- Children: Have smaller, developing kidneys and are more sensitive to invasive procedures.
- Adults: Renal artery injection can be used, whereas intravenous administration is generally preferred in pediatric patients.
- Outcome assessment criteria:
- Adults: Delay progression to end-stage renal disease (ESRD) or improvement in glomerular filtration rate (GFR).
- Children: In addition to renal function, long-term growth and developmental outcomes are also emphasized.
- Safety and ethical considerations:
- Adults: Clinical research has accumulated more extensive safety data.
- Children: Studies are still in early stages; therefore, greater caution is required regarding experimental therapies and invasive procedures.
Conflicting Results and Limitations of Current Clinical Trials
Current evidence regarding MSC therapy in pediatric kidney diseases remains preliminary. Larger clinical trials, standardized methodologies, and long-term follow-up are required to clearly determine the efficacy and safety of this therapeutic approach.
Challenges and Future Directions
Addressing challenges related to cell sources, delivery methods, mechanisms of action, biomarkers, and research standardization will help advance the development of personalized, safe, and more effective MSC therapies for children with kidney diseases.
Conclusion
Mesenchymal stem cell (MSC) therapy represents a promising therapeutic approach for pediatric kidney diseases due to its multiple mechanisms of action, including immunomodulation, tissue regeneration, anti-fibrotic effects, and paracrine signaling. Preclinical studies and early-phase clinical trials suggest that MSCs may support the treatment of various pediatric kidney disorders, such as acute kidney injury (AKI), chronic kidney disease (CKD), glomerular diseases, and congenital anomalies of the kidney and urinary tract (CAKUT).
However, several challenges must be addressed before widespread clinical application can be achieved. Therapeutic efficacy depends on the source of MSCs (bone marrow, umbilical cord, or adipose tissue) and the method of cell delivery. Intravenous administration is convenient but has limited renal homing efficiency. Furthermore, pediatric applications require consideration of unique factors such as disease etiology, organ development, treatment goals, and stricter safety requirements compared with adult patients.
Current limitations include methodological heterogeneity among studies, small sample sizes, variations in treatment protocols, and a lack of long-term safety data. Therefore, future research should focus on identifying biomarkers for treatment response, standardizing protocols, developing pediatric-specific research models, optimizing MSC delivery strategies, and establishing appropriate regulatory frameworks.
Collaboration among nephrologists, stem cell researchers, and regulatory authorities will play a crucial role in translating these promising research findings into safe and effective clinical therapies, ultimately improving the prognosis of children with kidney diseases.
References
Bahroudi, M., & Moghtaderi, M. (2026). Therapeutic Potential of Mesenchymal Stem Cells in Pediatric Kidney Disorders: A Comprehensive Review. Health science reports, 9(2), e71769.
Source: Health Science Reports
Link: https://onlinelibrary.wiley.com/doi/full/10.1002/hsr2.71769