Medical Express, May 1 , 2024
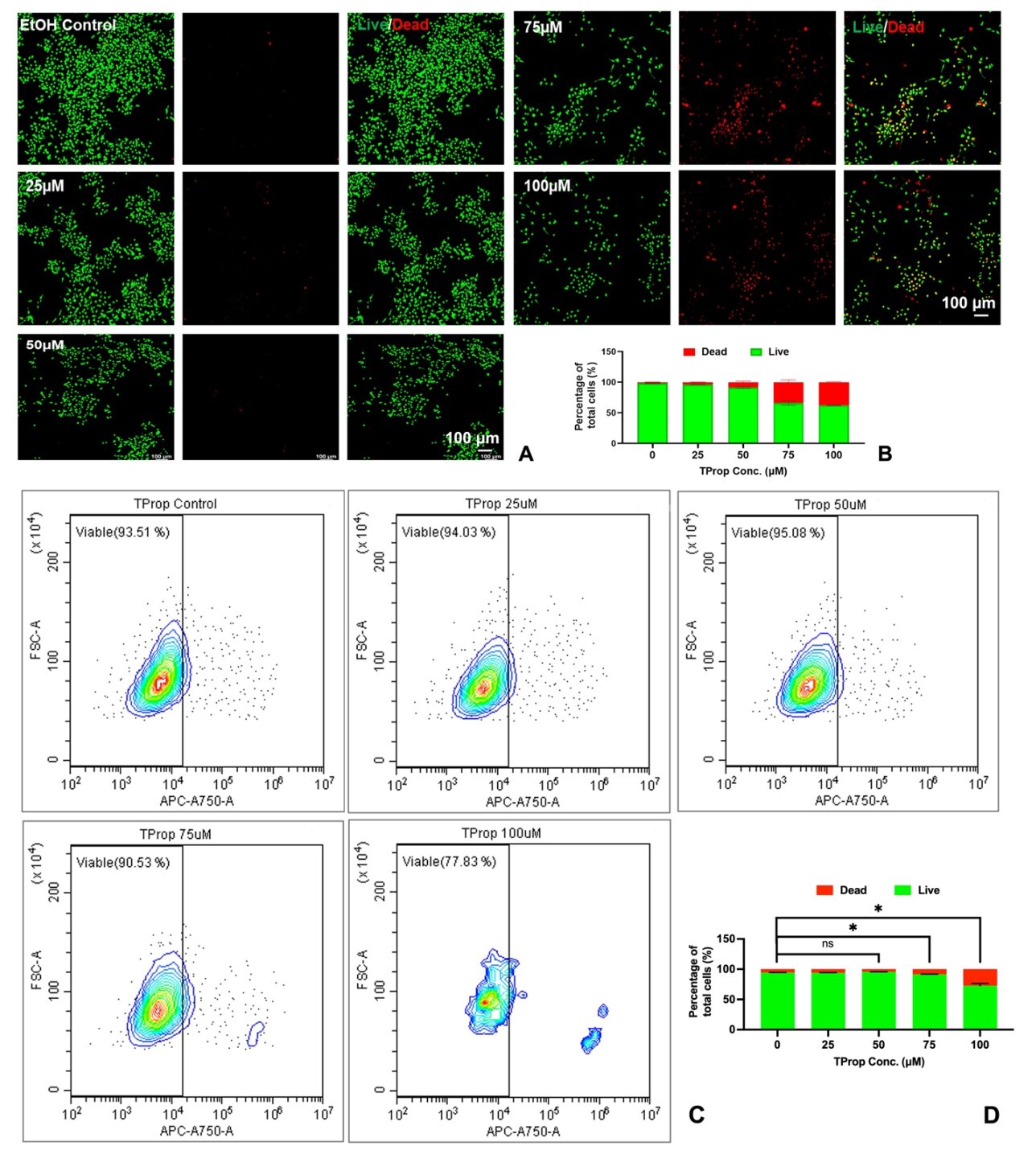
Live/dead assay to observe the cytotoxicity after neural stem cells (NSCs) incubation with TProp. Credit: Advanced Functional Materials (2023). DOI: 10.1002/adfm.202309866
Researchers at the University of Maryland School of Medicine (UMSOM) have identified an innovation in stem cell therapy to regenerate neural cells in the brain after cardiac arrest in an animal model. The study led by Xiaofeng Jia, BM, MS, Ph.D., FCCM, Professor of Neurosurgery, found that the application of modified sugar molecules on human neural stem cells improved the likelihood of the therapy’s success.
The application of these sugar molecules both enhanced the stem cells’ proliferation and their transition into neurons to help repair critical connections in the brain. The finding could eventually lead to improved recovery of patients with cardiac-arrest induced brain injuries.
The research is published in the journal Advanced Functional Materials.
Brain injury is the most common consequence of cardiac arrest, due to the impaired blood flow and oxygen to the brain. About 70% of the nearly 7 million people who suffer from cardiac arrest each year experience a long-term brain injury that leads to permanent disability.
The potential of stem cell therapy to address neurological dysfunction has long been fraught with challenges due to the harsh in vivo microenvironment of the brain; this results in poor stem cell retention and integration at the site of injuries.
Recent advances in manipulating a cell’s complex carbohydrate structure through metabolic glycoengineering, has enabled UMSOM researchers to explore the efficacy of a modified sugar molecule, known as the TProp sugar analog, to help stem cells remain more viable in the brain.
“All cells in a person’s body are enveloped in sugar molecules called ‘glycans,’“ said Dr. Jia. “Through our previous research, we were able to find that these sugar molecules are vital to cell function. Glycoengineering has enabled us to further enhance stem cell viability so they may provide therapeutic effects for cardiac-arrest-induced brain injuries. This is a very important step forward in regenerative medicine for patients.”
In the study, researchers examined the efficacy in a rat model and compared the effects of “naive” human neural stem cells to neural stem cells that were pretreated with the “TProp” sugar analog. The study found that stem cells pretreated with TProp, substantially improved brain function and reduced anxiety and depression-associated behaviors through various behavioral tests.
The treatment also activated the related inflammatory Wnt/β-catenin signaling pathway, which regulates critical aspects of cell function. This upregulated pathway by TProp promotes the transition of stem cells into neurons, the nerve cells responsible for sending and receiving signals from the brain.
The TProp-pretreated group also demonstrated improved synaptic plasticity, the ability of neurons to modify the strength of their connections, and reduced neuroinflammation in the central nervous system, providing a superior ability to regenerate and recover from damaged brain functions.
The results indicate that glycoengineered stem cells have the potential to promote the growth of new connections among surviving or regenerated neurons, leading to regenerated circuits in the brain.
“This innovative research has been an important proof of concept study suggesting that stem cells could be used to regenerate neural connections in the brain of patients who suffer a devasting injury after cardiac arrest,” said Dean Mark T. Gladwin, MD, who is the John Z. and Akiko K. Bowers Distinguished Professor and Dean, UMSOM, and Vice President for Medical Affairs, University of Maryland, Baltimore.
“Next steps for this translational application include determining the optimal delivery route and timing of metabolically glycoengineered stem cell therapy, as well as systemic evaluation on large animals before this can move into clinical studies.”
References
Jian Du et al, Metabolically Glycoengineered Neural Stem Cells Boost Neural Repair After Cardiac Arrest, Advanced Functional Materials (2023). DOI: 10.1002/adfm.202309866
Source: Medical Express
Link: https://medicalxpress.com/news/2024-05-preclinical-stem-cell-therapy-boosts.html