Open life sciences, 20/03/2026
Introduction
Pulmonary fibrosis is a group of lung disorders characterized by excessive accumulation of fibrous tissue, causing permanent scarring, reduced elasticity, and impaired respiratory function. Idiopathic Pulmonary Fibrosis (IPF) is the most common and severe form, primarily in older adults, associated with genetic, environmental, occupational factors, and certain drugs such as bleomycin, amiodarone, and methotrexate. Leukocyte activation and infiltration promote collagen deposition through proinflammatory cytokines and chemokines, indicating an important role of immunity in disease progression.
Mesenchymal Stem Cells (MSCs) from bone marrow (BM-MSCs) and umbilical cord (UC-MSCs) have differentiation and immunomodulatory capabilities, improving lung morphology and respiratory function in IPF models via antifibrotic and anti-inflammatory effects. However, studies comparing the efficacy and mechanisms of the two types of MSCs are limited. Investigating the potential of BM-MSCs and UC-MSCs is necessary to optimize IPF treatment strategies.
Materials and methods
Animal model and housing conditions
The study used 7–8-week-old male BALB/c mice, housed in a pathogen-free environment with a temperature of 23–25 °C, stable humidity, a balanced diet, and ad libitum access to water. Experiments, including bleomycin-induced pulmonary fibrosis, were conducted in accordance with ethical guidelines and approved by the Institutional Animal Ethics Committee of Chung Shan Medical University (Approval No. 2429).
Animal experimental design
Preparation and characterization of MSCs
In this study, the primary aim was to evaluate the therapeutic efficacy of UC-MSCs and BM-MSCs in bleomycin-induced pulmonary fibrosis mice. UC-MSCs were isolated from the umbilical cords of healthy 30-year-old women with consent. Umbilical cords were processed within 24 hours after birth: washed with DPBS, sterilized with 75 % ethanol, blood vessels removed, and Wharton’s jelly minced into 0.5–1 mm³ pieces. Samples were cultured in α-MEM with 5 % UltraGRO™ and antibiotics at 37 °C, 5 % CO2, with medium changed every 3–4 days. Upon reaching confluency, cells were detached using TrypLE™ Express and expanded. Passage 3 (P3) cells were cryopreserved in CryoStor CS10 at −190 °C. BM-MSCs were obtained from the bone marrow of healthy donors (24–42 years old) via iliac crest aspiration. Mononuclear cells were isolated using Ficoll-Paque and cultured in α-MEM with 10 % FBS and antibiotics; non-adherent cells were removed after 48–72 h, and adherent MSCs were expanded and used at P3.
Donor selection criteria included absence of chronic inflammatory, autoimmune, infectious, or malignant diseases; exclusion of individuals with systemic illness, long-term medication use, or abnormal laboratory results.
Animal experimental design and treatment
A total of 24 male BALB/c mice were randomly divided into 4 groups:
- Control group (CON): Intratracheal injection of saline on days 0 and 14.
- Bleomycin group (BLE): Intratracheal injection of bleomycin 5 mg/kg on day 0 and saline on day 14.
- UC-MSC group: Intratracheal injection of bleomycin on day 0, followed by 1 × 10⁶ UC-MSCs on day 14.
- BM-MSC group: Intratracheal injection of bleomycin on day 0, followed by 1 × 10⁶ BM-MSCs on day 14.
Mice were monitored daily and euthanized early if body weight decreased by >15 %, showed poor feeding, or exhibited signs of stress. On day 42, all mice were euthanized to collect lung tissue and bronchoalveolar lavage fluid (BALF) for fibrosis, inflammation, and leukocyte infiltration analyses; BALF was centrifuged, with the supernatant stored at −80 °C for cytokine measurement, and the cell pellet used for leukocyte profiling by flow cytometry.
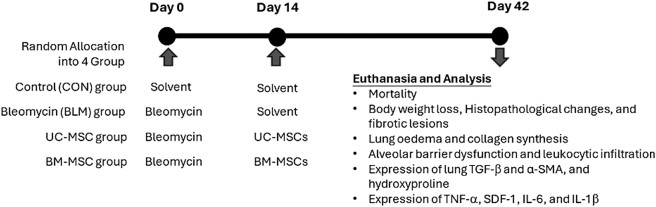
Figure 1. Flowchart of experimental procedures applied to bleomycin-induced IPF animal model treated with UC-MSCs or BM-MSCs.
Animal mortality monitoring
After bleomycin injection to induce pulmonary fibrosis, mouse mortality was monitored over 42 days. Physiological parameters, including heart rate, respiration, eye reflexes, and overall body movements, were assessed to determine mortality status.
Lung tissue weight and pulmonary index analysis
After euthanasia, mouse lung tissues were excised and weighed for wet weight. The tissues were then dried at 80 °C for 48 hours to determine dry weight. Pulmonary edema was quantitatively assessed by the wet/dry ratio (wet weight divided by dry weight). The lung index, reflecting lung enlargement relative to body weight, was calculated as wet lung weight (mg) divided by body weight (g).
Lung tissue preparation
On day 42, lung tissues were excised and fixed in 10 % formalin, embedded in paraffin, and sectioned for microscopic analysis. Sections were stained with H&E or Masson’s trichrome to assess cellular structure and fibrotic tissue. Fibrosis was quantified using the Ashcroft score (0–8) and the percentage of fibrotic area relative to the total field area (%), allowing comparison of fibrosis severity between groups.
Immunohistochemistry analysis
Lung sections were deparaffinized and rehydrated for staining. Antigen retrieval was performed using EDTA solution, and hydrogen peroxide was applied to reduce nonspecific staining. After blocking, sections were incubated with primary antibodies recognizing TNFα and α-SMA (a biomarker of smooth muscle cells), followed by incubation with a peroxidase-conjugated secondary antibody. Diaminobenzidine was used as the chromogen, producing a brown precipitate at antigen expression sites. Sections were then analyzed using the Lion Heart FX automated microscope.
Enzyme-linked immunosorbent assay
ELISA was performed to quantify collagen, hydroxyproline, SDF-1, and TGF-β in lung tissue samples. Samples were frozen, homogenized, and centrifuged at 5,000×g for 10 minutes at 4 °C to collect the supernatant, which was then used in the corresponding ELISA kits. Additionally, bronchoalveolar lavage fluid (BALF) was analyzed by ELISA to measure TNF-α, IL-1β, and IL-6.
Leukocyte infiltration in BALF
Leukocyte infiltration in BALF was assessed by flow cytometry. After centrifugation, the cell pellet was resuspended in PBS containing 1 % bovine serum albumin. For immunophenotyping, cells were incubated with fluorochrome-conjugated antibodies recognizing surface markers: CD45 (total leukocytes), CD11b (myeloid cells), Ly6G (neutrophils), and CD3 (T cells). Leukocytes were identified as CD45⁺, granulocytes as CD45⁺CD11b⁺, neutrophils as CD45⁺Ly6G⁺, and lymphocytes as CD45⁺CD3⁺. Flow cytometry analysis included unstained and isotype controls to determine background signal and set gating. Stained cells were resuspended in PBS and analyzed using an Accuri C6 cytometer with accompanying software.
Statistical analysis
Statistical analysis was performed using SPSS. Data are presented as mean ± SD. Normality was assessed by the Shapiro-Wilk test; ANOVA with Bonferroni post hoc was used for normally distributed data, and Kruskal-Wallis with Dunn’s post hoc for non-normal data. Statistical significance: compared to CON # P < 0.05; compared to BLE *P < 0.05; compared to UC-MSC $ P < 0.05.
Results
Alleviation of mortality in bleomycin-induced IPF by UC-MSCs and BM-MSCs
To evaluate the efficacy of UC-MSCs and BM-MSCs in reducing bleomycin-induced pulmonary fibrosis mortality, mice were treated with MSCs on day 14 after bleomycin injection. Mortality was recorded daily over 42 days. The BLE group showed significantly higher mortality compared to the CON group, while UC-MSCs reduced mortality more effectively than BM-MSCs.
Effect of UC-MSCs and BM-MSCs on body weight loss, histopathological changes, and fibrotic lesions
Results showed that bleomycin caused a significant body weight loss (P < 0.05). Treatment with UC-MSCs, but not BM-MSCs, significantly attenuated bleomycin-induced weight loss. Histopathological changes assessed by H&E staining revealed structural lung damage, thickened alveolar walls, and pronounced leukocyte infiltration in the BLE group compared to the CON group. Both UC-MSCs and BM-MSCs significantly improved histological changes and Ashcroft scores (P < 0.05). Fibrotic areas, assessed by Masson’s trichrome staining, were greater in the BLE group; both MSCs significantly reduced fibrosis (P < 0.05), with UC-MSCs showing superior efficacy in reducing fibrosis and restoring lung structure.
Effect of UC-MSCs and BM-MSCs on lung edema and collagen synthesis
The lung index and wet/dry ratio are key indicators for assessing pulmonary edema in the IPF model. Bleomycin caused significant pulmonary edema, which was markedly improved after treatment with UC-MSCs or BM-MSCs (P < 0.05). Collagen expression increased significantly after bleomycin and was markedly reduced by UC-MSC treatment, whereas BM-MSCs did not produce a similar effect (P < 0.05).
Alleviation of alveolar barrier dysfunction and leukocytic infiltration following UC-MSC and BM-MSC intervention
Alveolar barrier dysfunction was assessed based on protein concentration in BALF. Bleomycin caused significant alveolar barrier disruption (P < 0.05), which was markedly improved by UC-MSC treatment but not to the same extent by BM-MSCs. This dysfunction allowed leukocytes, including granulocytes, neutrophils, and lymphocytes, to infiltrate lung tissue, promoting inflammatory responses. Leukocyte, granulocyte, neutrophil, and lymphocyte infiltration increased significantly after bleomycin (P < 0.05), but treatment with either UC-MSCs or BM-MSCs significantly alleviated leukocyte infiltration in fibrotic lesions (P < 0.05).
Reduced expression of lung TGF-β, a-SMA, and hydroxyproline via UC-MSCs
In the progression of pulmonary fibrosis, the BLE group showed increased expression of profibrotic markers TGF-β and α-SMA, consistent with advancing fibrosis. Immunohistochemical analysis demonstrated that UC-MSCs markedly reduced TGF-β and α-SMA, whereas BM-MSCs showed only limited reduction. ELISA analysis revealed that both MSCs significantly decreased TGF-β in lung tissue (P < 0.05), but only UC-MSCs significantly reduced hydroxyproline (P < 0.05), a marker of collagen accumulation. These results highlight the superior antifibrotic efficacy of UC-MSCs, inhibiting TGF-β, reducing collagen deposition, and suppressing myofibroblast activity, while BM-MSCs provided limited improvement.
Effect of UC-MSCs and BM-MSCs on TNF-a, SDF-1, IL-6, and IL-1β expression
ELISA analysis showed that TNF-α, SDF-1, IL-6, and IL-1β in BALF were significantly increased in the BLE group compared to CON (P < 0.05). Both UC-MSCs and BM-MSCs significantly reduced TNF-α (P < 0.05), whereas only UC-MSCs significantly decreased SDF-1, IL-6, and IL-1β (P < 0.05).
Discussion
- IPF and Research Model
- IPF is a progressive lung disease characterized by fibrosis, dyspnea, dry cough, and fatigue; it mainly affects individuals aged 70–75 years, with a median survival of 2–5 years after diagnosis.
- The etiology is unclear, disease progression is heterogeneous, and effective treatment options are limited.
- The bleomycin-induced mouse model reproduces key features of IPF: weight loss, pulmonary edema, fibrotic lesions, increased collagen, and histopathological changes.
- Efficacy of UC-MSCs and BM-MSCs
- Mortality: Both UC-MSCs and BM-MSCs reduced mortality compared to the BLE group, with UC-MSCs being more effective.
- Body weight & lung structure: UC-MSCs better restored body weight and lung structure than BM-MSCs.
- Fibrosis & histology: UC-MSCs significantly reduced fibrotic lesions, collagen, TGF-β, α-SMA, and hydroxyproline; BM-MSCs reduced TGF-β and α-SMA but did not decrease hydroxyproline.
- Alveolar barrier & inflammation: UC-MSCs more effectively improved alveolar barrier function, reduced leukocyte infiltration, and decreased proinflammatory cytokines (TNF-α, SDF-1, IL-6, IL-1β) compared to BM-MSCs.
- Mechanisms & Advantages of UC-MSCs
- High proliferative capacity, low immunogenicity, and genetic stability.
- Modulate cytokines, reduce TGF-β, NF-κB, and Wnt/β-catenin signaling → decrease fibrosis and inflammation.
- BM-MSCs have more limited effects, particularly on hydroxyproline and lung structure regeneration.
- Study Limitations
- The bleomycin model does not fully recapitulate human IPF.
- Only a single dose and schedule were tested; long-term cell distribution, persistence, and regulatory cytokines such as IL-10 were not evaluated.
- Further studies are needed to confirm the clinical efficacy of UC-MSCs and BM-MSCs.
Conclusion
This study confirms that the pathological features of bleomycin-induced IPF, including mortality, weight loss, and pulmonary fibrosis, can be reproduced in mice. Administration of UC-MSCs significantly reduced mortality, weight loss, histopathological damage, fibrotic injury, collagen accumulation, and expression of TGF-β, α-SMA, and hydroxyproline, while also improving pulmonary edema, leukocyte infiltration, alveolar barrier dysfunction, and proinflammatory cytokines. BM-MSCs provided only partial improvement, mainly reducing pulmonary edema, leukocyte infiltration, and TNF-α expression, but were less effective than UC-MSCs. These results indicate that UC-MSCs have superior therapeutic efficacy compared to BM-MSCs in bleomycin-induced IPF.
References
Ni, Y. L., Shen, H. T., Lee, C. Y., Lee, T. P., Sieber, M., Tseng, C. C., Tsao, C. T., & Kuan, Y. H. (2026). UC-MSCs exhibit superior antifibrotic and anti-inflammatory effects compared to BM-MSCs in a bleomycin-induced idiopathic pulmonary fibrosis model. Open life sciences, 21(1), 20251293.
Source: Open life sciences